Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000546.5 | RefSeq Select | 2591 nt | 203–1384 |
| NM_000546.3 | Alternative | 2640 nt | 252–1433 |
| NM_000546.6 | MANE Select | 2512 nt | 143–1324 |
| NM_000546.4 | Alternative | 2586 nt | 198–1379 |
| NM_000546.2 | Alternative | 2629 nt | 252–1433 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThis sequence change replaces leucine, which is neutral and non-polar, with arginine, which is basic and polar, at codon 265 of the TP53 protein (p.Leu265Arg). This variant is not present in population databases (gnomAD no frequency). This variant has not been reported in the literature in individuals affected with TP53-related conditions. Advanced modeling performed at Invitae incorporating data from internal and/or published experimental studies (PMID: 12826609, 29979965, 30224644) indicates that this missense variant is expected to disrupt TP53 function. In summary, the available evidence is currently insufficient to determine the role of this variant in disease. Therefore, it has been classified as a Variant of Uncertain Significance.
"This variant has been reported in ClinVar as Uncertain significance (1 clinical laboratories)."
COSMIC Somatic Evidence
Open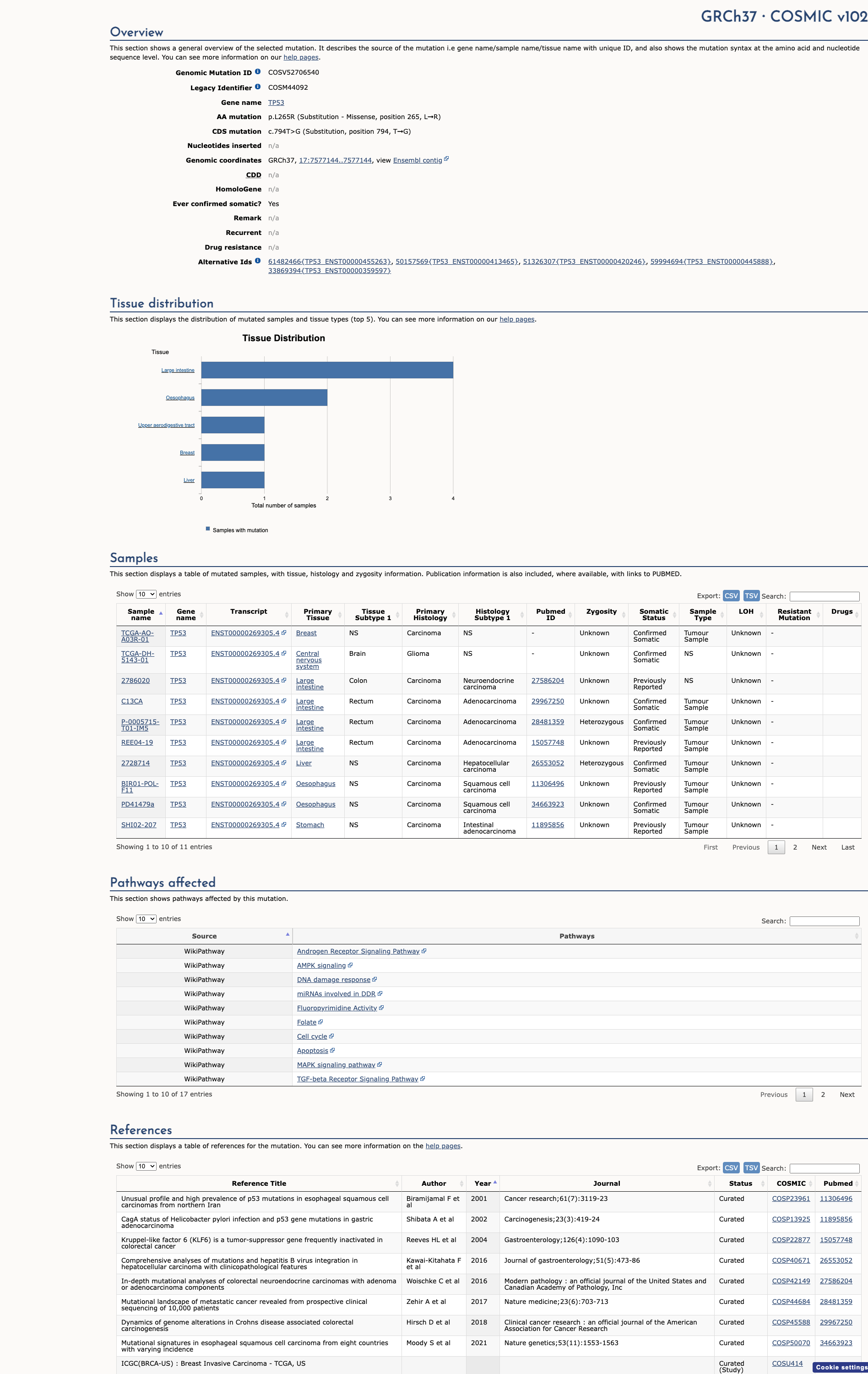
Functional Impact & Domains
Functional Domain
The TP53 L265R variant has been functionally characterized and demonstrated to have a damaging effect. In vivo studies with yeast and in vitro studies with human cancer cell lines have shown that this mutation is inactivating, resulting in a loss of transactivational activity and reduced growth suppression activity compared to the wildtype protein.
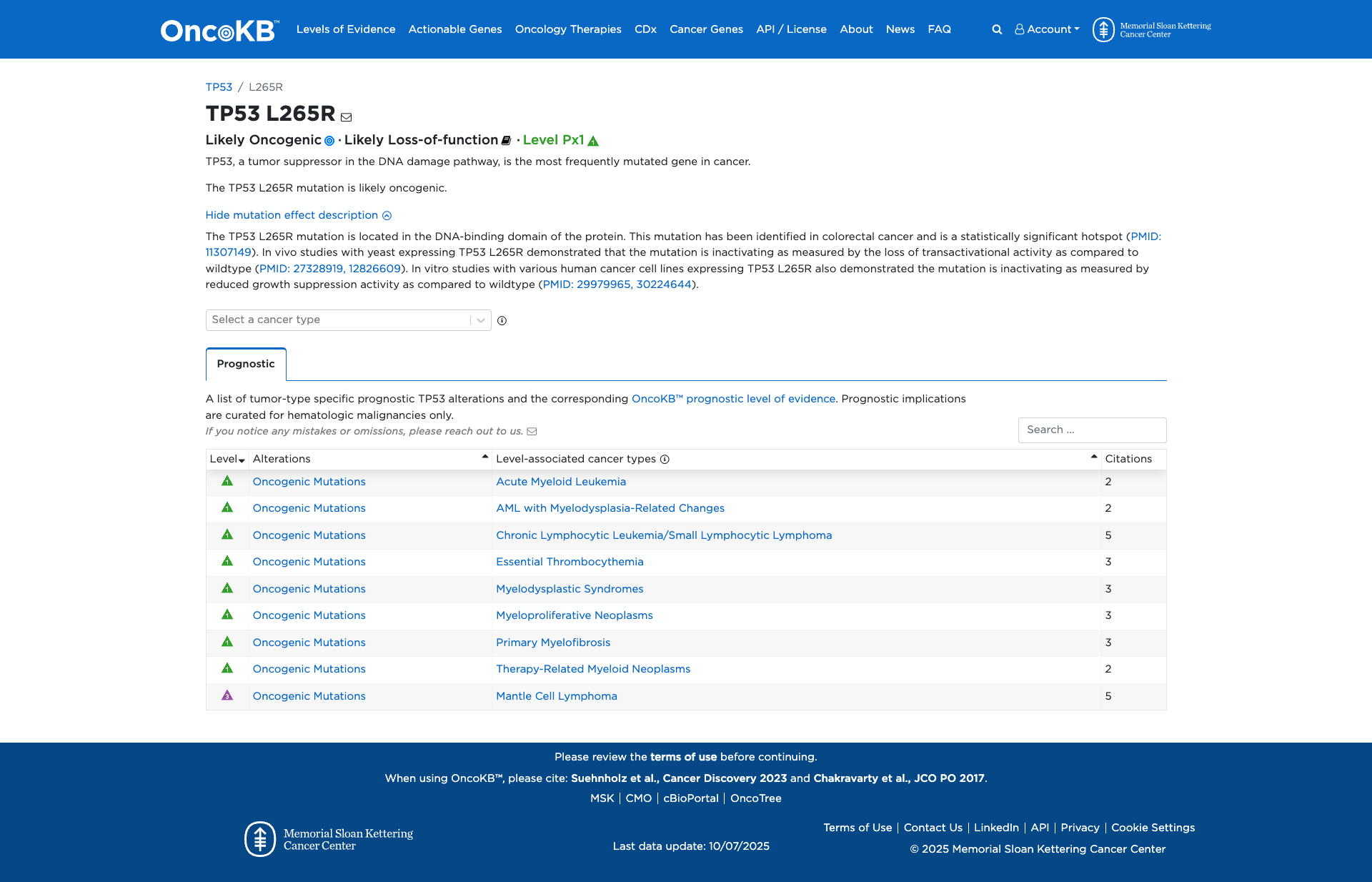
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.02 | -13 bp |
| Donor Loss (DL) | 0.0 | 65 bp |
| Acceptor Gain (AG) | 0.0 | 14 bp |
| Donor Gain (DG) | 0.0 | -19 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Not Applied)
According to VCEP guidelines, PVS1 applies to null variants causing NMD or affecting canonical splice sites. The variant is a missense change (L265R), not a null variant. Therefore, PVS1 is not applied.
PS1 (Not Applied)
According to VCEP guidelines, PS1 applies when a variant results in the same amino acid change as a previously established pathogenic variant. L265R is novel with no prior TP53 variants at codon 265 classified as Pathogenic. Therefore, PS1 is not applied.
PS2 (Not Applied)
According to VCEP guidelines, PS2 is based on confirmed de novo occurrence. No de novo data are available for this variant. Therefore, PS2 is not applied.
PS3 (Not Applied)
According to VCEP guidelines, PS3_Strong requires non‐functional in Kato et al. data AND LOF in another assay, and PS3_Supporting requires specific Kotler and Giacomelli data if Kato data are absent. The functional evidence provided (yeast and cancer cell line assays) cannot be mapped to the VCEP‐specified assay series. Therefore, PS3 is not applied.
PS4 (Not Applied)
According to VCEP guidelines, PS4 is based on proband counts/points. No case series or proband point data are available. Therefore, PS4 is not applied.
PM1 (Not Applied)
According to VCEP guidelines, PM1_Moderate applies to missense variants at codons 175, 245, 248, 249, 273, or 282, or hotspots with ≥10 somatic occurrences. L265 is outside these codons and has zero COSMIC occurrences. Therefore, PM1 is not applied.
PM2 (Supporting)
According to VCEP guidelines, PM2_Supporting applies when allele frequency is <0.00003 in gnomAD. The variant is absent from gnomAD (allele frequency = 0). Therefore, PM2 is applied at Supporting strength.
PM3 (Not Applied)
According to standard ACMG guidelines, PM3 applies to recessive disorders with variants in trans. TP53‐related disease is autosomal dominant and no trans observations are present. Therefore, PM3 is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, PM4 applies to protein length changes (in‐frame indels, stop‐loss). L265R is a missense change. Therefore, PM4 is not applied.
PM5 (Not Applied)
According to VCEP guidelines, PM5 applies when other pathogenic missense changes at the same residue exist. No other TP53 variants at codon 265 have been classified as Pathogenic. Therefore, PM5 is not applied.
PM6 (Not Applied)
According to VCEP guidelines, PM6 applies to assumed de novo variants without confirmation. No de novo information is available. Therefore, PM6 is not applied.
PP1 (Not Applied)
According to VCEP guidelines, PP1 requires segregation data (3–7 meioses). No family segregation data are available. Therefore, PP1 is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, PP2 applies when missense variants in a gene with low benign variation are observed. TP53 has a high rate of pathogenic missense variants and benign variation is not notably low. Therefore, PP2 is not applied.
PP3 (Not Applied)
According to VCEP guidelines, PP3 requires specific BayesDel and aGVGD metrics. Only REVEL score is provided. Without VCEP‐specified metrics, PP3 cannot be applied. Therefore, PP3 is not applied.
PP4 (Not Applied)
According to VCEP guidelines, PP4 applies to variant observations with specific VAF distributions in tumor data. No such data are provided. Therefore, PP4 is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, PP5 applies to reputable sources reporting pathogenicity. ClinVar lists this variant as VUS. Therefore, PP5 is not applied.
BA1 (Not Applied)
According to VCEP guidelines, BA1 applies when allele frequency ≥0.001. The variant is absent from gnomAD. Therefore, BA1 is not applied.
BS1 (Not Applied)
According to VCEP guidelines, BS1 applies when allele frequency ≥0.0003 but <0.001. The variant is absent from gnomAD. Therefore, BS1 is not applied.
BS2 (Not Applied)
According to VCEP guidelines, BS2 requires observation in multiple older cancer‐free females. No such data exist. Therefore, BS2 is not applied.
BS3 (Not Applied)
According to VCEP guidelines, BS3 applies when functional assays show preserved function. Available studies show loss of function, not preserved function. Therefore, BS3 is not applied.
BS4 (Not Applied)
According to VCEP guidelines, BS4 requires lack of segregation with disease. No segregation data are available. Therefore, BS4 is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, BP1 applies to missense in genes where only truncating variants cause disease. TP53 disease is driven by missense variants. Therefore, BP1 is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, BP2 applies when observed in trans with a pathogenic variant for a dominant gene. No such observations are available. Therefore, BP2 is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, BP3 applies to in‐frame indels in repetitive regions. This is a missense variant. Therefore, BP3 is not applied.
BP4 (Not Applied)
According to VCEP guidelines, BP4 requires specific BayesDel thresholds and absence of splice impact. Only REVEL data are provided. Therefore, BP4 is not applied.
BP5 (Not Applied)
According to standard ACMG guidelines, BP5 applies when a variant co‐occurs with another pathogenic variant. No co‐occurrence data are available. Therefore, BP5 is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, BP6 applies to unpublished reputable source assertions. No such assertions exist. Therefore, BP6 is not applied.
BP7 (Not Applied)
According to VCEP guidelines, BP7 applies to silent or intronic variants outside core splice sites with no splice impact. This is a missense variant. Therefore, BP7 is not applied.