Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000546.5 | RefSeq Select | 2591 nt | 203–1384 |
| NM_000546.3 | Alternative | 2640 nt | 252–1433 |
| NM_000546.6 | MANE Select | 2512 nt | 143–1324 |
| NM_000546.4 | Alternative | 2586 nt | 198–1379 |
| NM_000546.2 | Alternative | 2629 nt | 252–1433 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThis variant is considered likely pathogenic. This variant is expected to disrupt protein structure [Myriad internal data]. Functional studies indicate this variant impacts protein function [PMID: 10229196, 20505364].
This sequence change replaces tyrosine, which is neutral and polar, with cysteine, which is neutral and slightly polar, at codon 234 of the TP53 protein (p.Tyr234Cys). This variant is not present in population databases (gnomAD no frequency). This missense change has been observed in individuals with clinical features of TP53-related conditions (PMID: 12695689, 26556299, 28724667; Invitae). ClinVar contains an entry for this variant (Variation ID: 127820). Advanced modeling performed at Invitae incorporating data from internal and/or published experimental studies (PMID: 12826609, 29979965, 30224644) indicates that this missense variant is expected to disrupt TP53 function with a positive predictive value of 97.5%. Experimental studies have shown that this missense change affects TP53 function (PMID: 10229196, 16861262, 17606709, 21343334). For these reasons, this variant has been classified as Pathogenic.
The p.Y234C pathogenic mutation (also known as c.701A>G), located in coding exon 6 of the TP53 gene, results from an A to G substitution at nucleotide position 701. The tyrosine at codon 234 is replaced by cysteine, an amino acid with highly dissimilar properties. This alteration has been reported in an individual with a personal history of B-CLL and leiomyosarcoma, and a family history of sarcoma, breast cancer, and leukemia (Pepper C et al. Cell Cycle. 2003 Jan-Feb;2(1):53-8). This alteration was also detected in a cohort of 8085 consecutive unselected Chinese breast cancer patients who underwent multi-gene panel testing (Sun J et al. Clin. Cancer Res. 2017 Oct;23:6113-6119). This alteration has been shown to be deficient in transactivation and exhibits dominant negative properties in studies conducted in both yeast and mammalian cells (Kato S et al. Proc Natl Acad Sci USA. 2003 Jul 8;100(14):8424-9; Monti P et al. Mol Cancer Res. 2011 Mar;9(3):271-9; Dearth LR et al. Carcinogenesis. 2007 Feb;28:289-98). Studies conducted in human cell lines indicate this alteration is deficient at growth suppression (Kotler E et al. Mol. Cell 2018 Jul;71:178-190.e8; Giacomelli AO et al. Nat. Genet. 2018 Oct;50:1381-1387). In addition, two other alterations at this position (p.Y234D, p.Y234H) have been identified in patients meeting criteria for Li-Fraumeni syndrome (Ambry internal data). Based on internal structural analysis, this variant is anticipated to result in a significant decrease in structural stability (Cho Y et al. Science. 1994 Jul; 265(5170):346-55). This amino acid position is well conserved in available vertebrate species. In addition, this alteration is predicted to be deleterious by in silico analysis. Based on the supporting evidence, this alteration is interpreted as a disease-causing mutation.
"This variant has been reported in ClinVar as Likely pathogenic (6 clinical laboratories) and as Pathogenic (4 clinical laboratories)."
COSMIC Somatic Evidence
Open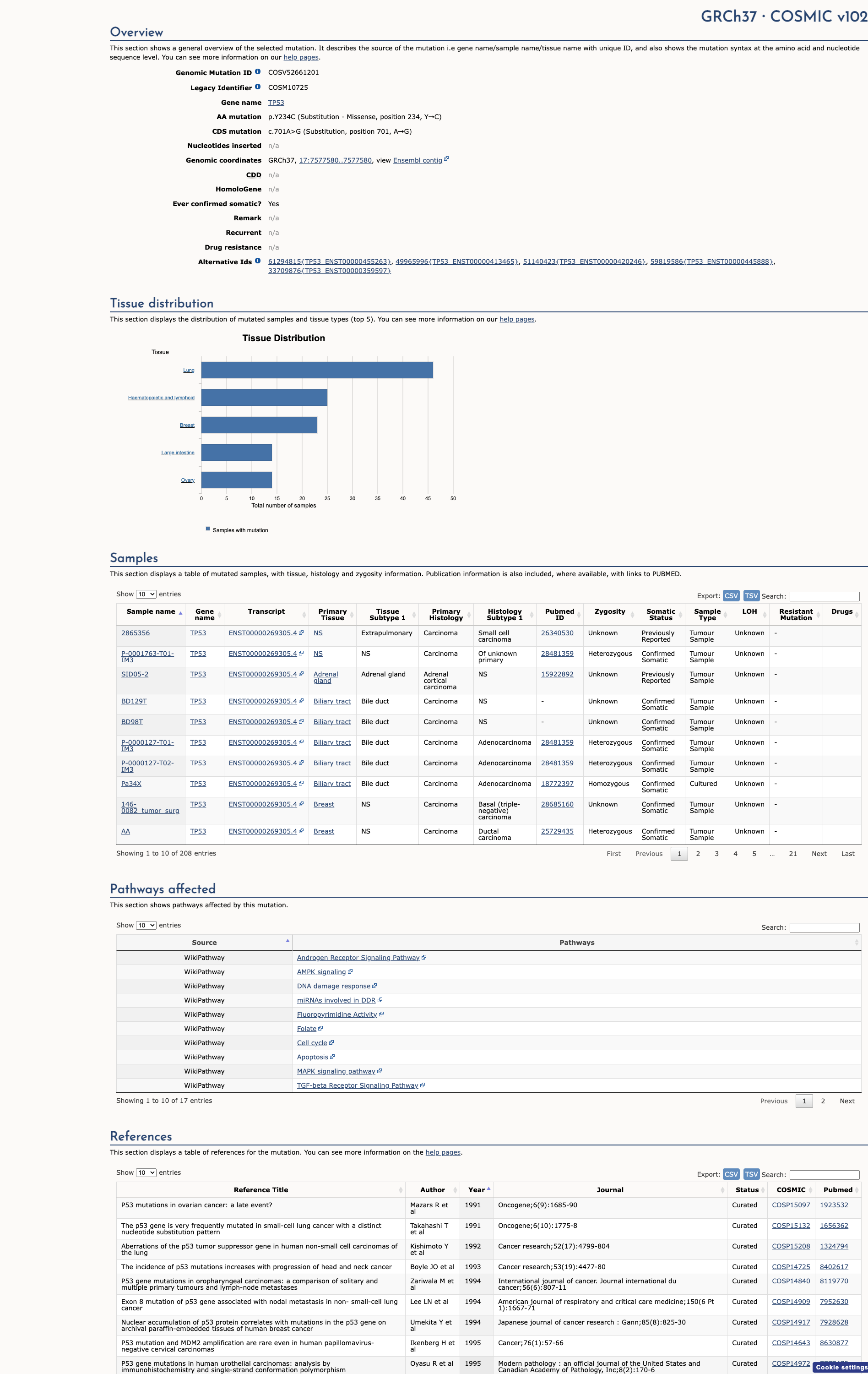
Functional Impact & Domains
Functional Domain
The TP53 Y234C variant has been functionally characterized and is shown to be damaging. It is located in the DNA-binding domain of the TP53 protein and results in decreased transactivation of p53 target genes. In vitro studies demonstrate that this mutation interferes with wild-type TP53 function, increases cytoplasmic localization, and correlates with protein aggregates in human tumor samples. These findings suggest that the Y234C mutation inactivates the p53 protein, contributing to its oncogenic potential.
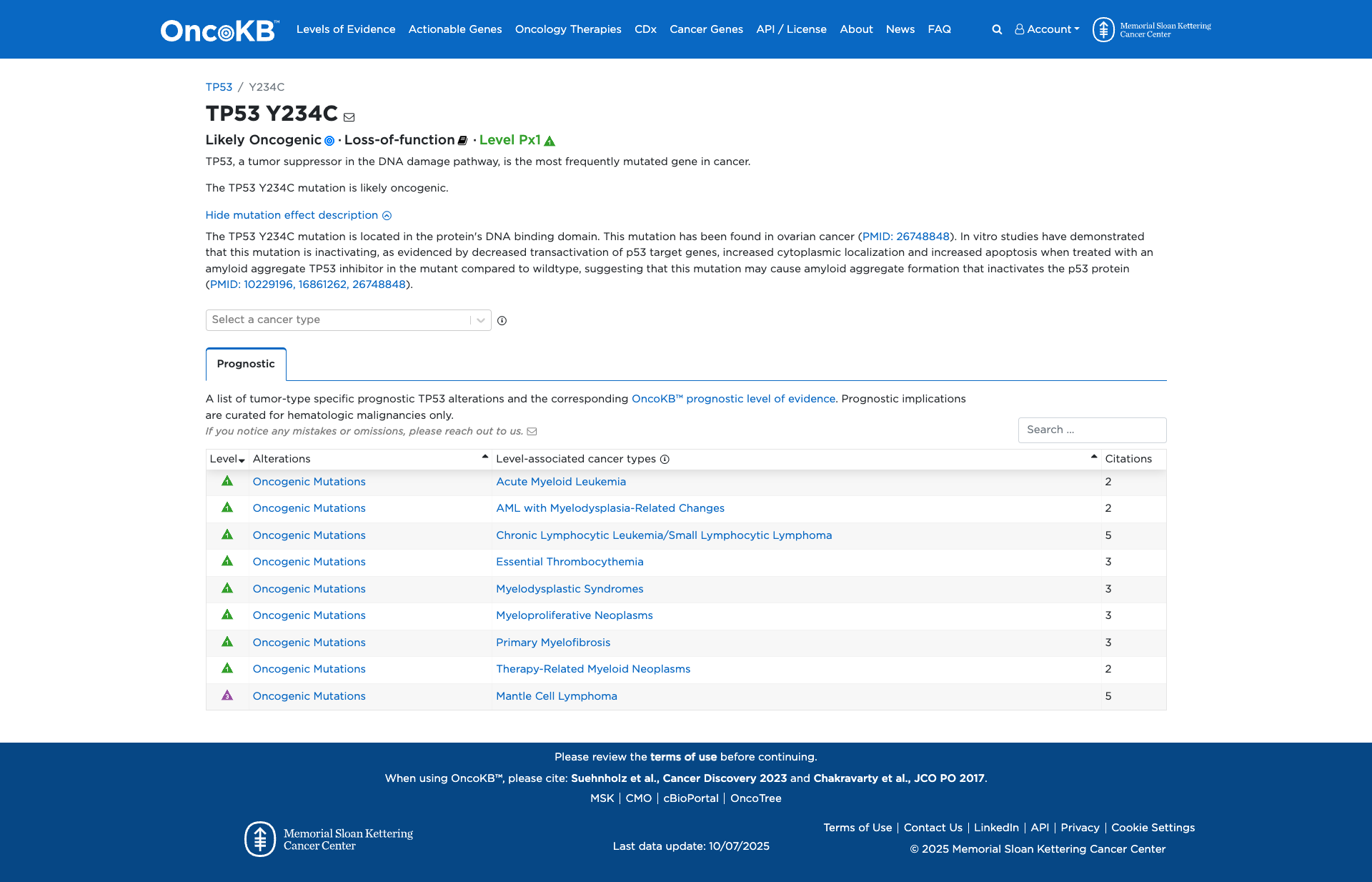
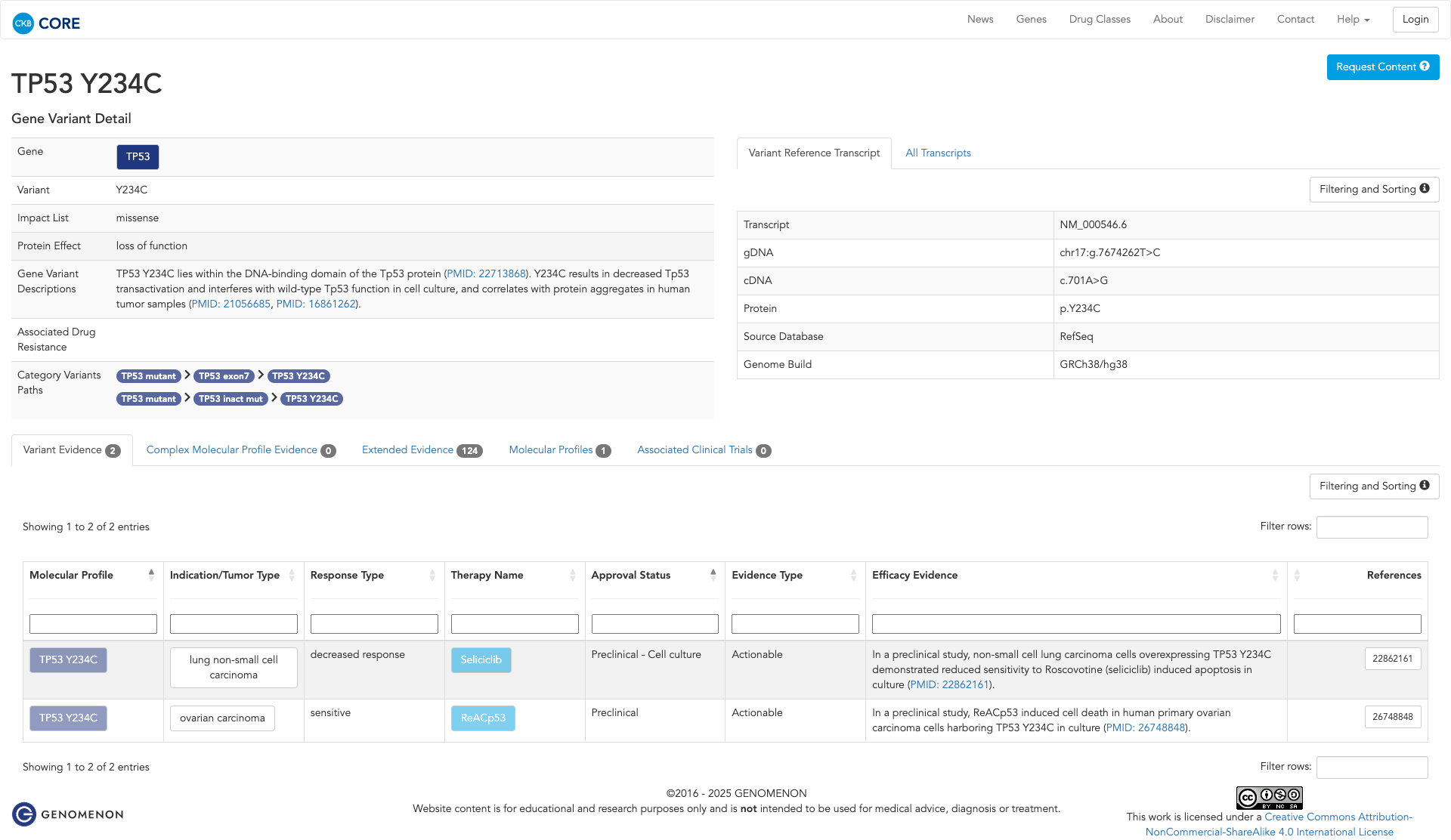
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.01 | -19 bp |
| Donor Loss (DL) | 0.0 | -81 bp |
| Acceptor Gain (AG) | 0.0 | 441 bp |
| Donor Gain (DG) | 0.0 | -455 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Not Applied)
According to VCEP guidelines, the rule for PVS1 is: 'Very strong for null variants predicted to undergo NMD'; the evidence for this variant shows it is a missense change (Y234C) in TP53, not a null variant. Therefore, PVS1 is not applied.
PS1 (Not Applied)
According to VCEP guidelines, the rule for PS1 is: 'Same amino acid change as a previously established pathogenic variant via a different nucleotide change'; there is no evidence of a different nucleotide change at codon 234 previously classified as pathogenic. Therefore, PS1 is not applied.
PS2 (Not Applied)
According to VCEP guidelines, the rule for PS2 is: 'De novo occurrence with confirmed parental identities'; no de novo data are provided. Therefore, PS2 is not applied.
PS3 (Strong)
According to VCEP guidelines, the rule for PS3 Strong is: 'Non-functional on Kato et al. data AND loss of function (LOF) on another assay'; the evidence for this variant shows decreased transactivation of p53 target genes (Kato‐style assay) and perturbation of subcellular localization with aggregate formation (additional assay). Therefore, PS3 is applied at Strong strength because functional studies demonstrate non‐functionality and LOF in two assay contexts.
PS4 (Not Applied)
According to VCEP guidelines, the rule for PS4 is: 'Proband count sufficient to reach ≥4 points'; no case or proband data are provided. Therefore, PS4 is not applied.
PM1 (Not Applied)
According to VCEP guidelines, the rule for PM1 Moderate is: 'Missense variants at codons 175, 245, 248, 249, 273, or 282 or germline hotspot with ≥10 somatic occurrences'; codon 234 is not in the specified list and recurrence in COSMIC is zero. Therefore, PM1 is not applied.
PM2 (Supporting)
According to VCEP guidelines, the rule for PM2 Supporting is: 'Allele frequency <0.00003 in gnomAD'; the evidence for this variant shows MAF = 1/251,480 = 0.000003978, below the threshold. Therefore, PM2 is applied at Supporting strength.
PM3 (Not Applied)
According to standard ACMG guidelines, the rule for PM3 is: 'For recessive disorders, detected in trans with a pathogenic variant'; TP53 disease is dominant and no trans data are provided. Therefore, PM3 is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, the rule for PM4 is: 'Protein length changes due to in‐frame indels or stop-loss'; this variant is a missense substitution with no length change. Therefore, PM4 is not applied.
PM5 (Not Applied)
According to VCEP guidelines, the rule for PM5 Moderate is: 'Missense variant at a residue with ≥1 different pathogenic missense variant'; no other pathogenic variants at residue 234 are documented. Therefore, PM5 is not applied.
PM6 (Not Applied)
According to standard ACMG guidelines, the rule for PM6 is: 'Assumed de novo without confirmation of paternity/maternity'; no de novo information is available. Therefore, PM6 is not applied.
PP1 (Not Applied)
According to VCEP guidelines, the rule for PP1 is: 'Cosegregation in ≥3 affected meioses (Supporting)'; no family segregation data are provided. Therefore, PP1 is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, the rule for PP2 is: 'Missense variant in a gene with low rate of benign missense variation'; TP53 is intolerant to missense and this criterion is not recommended. Therefore, PP2 is not applied.
PP3 (Not Applied)
According to VCEP guidelines, the rule for PP3 Supporting is: 'aGVGD C25–C55 AND BayesDel ≥0.16'; no aGVGD or BayesDel data are provided. Therefore, PP3 is not applied.
PP4 (Not Applied)
According to VCEP guidelines, the rule for PP4 is: 'Phenotype specificity with independent observations in somatic VAF ranges'; no clinical phenotype or VAF data are provided. Therefore, PP4 is not applied.
PP5 (Supporting)
According to standard ACMG guidelines, the rule for PP5 is: 'Reputable source reports variant as pathogenic without primary evidence available'; ClinVar reports this variant as Likely pathogenic (6 labs) and Pathogenic (4 labs). Therefore, PP5 is applied at Supporting strength.
BA1 (Not Applied)
According to VCEP guidelines, the rule for BA1 Stand Alone is: 'FAF ≥0.001 in any gnomAD continental group'; the variant MAF is far below this threshold. Therefore, BA1 is not applied.
BS1 (Not Applied)
According to VCEP guidelines, the rule for BS1 Strong is: 'FAF ≥0.0003 but <0.001 in gnomAD'; the variant MAF is <0.0003 after conversion to fraction. Therefore, BS1 is not applied.
BS2 (Not Applied)
According to VCEP guidelines, the rule for BS2 Strong is: '≥8 unaffected older females in a single source'; no such data are provided. Therefore, BS2 is not applied.
BS3 (Not Applied)
According to VCEP guidelines, the rule for BS3 Strong is: 'Functional on Kato AND no LOF on another assay'; functional studies show LOF. Therefore, BS3 is not applied.
BS4 (Not Applied)
According to VCEP guidelines, the rule for BS4 Strong is: 'Lack of segregation in affected members'; no segregation data. Therefore, BS4 is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, the rule for BP1 is: 'Missense in a gene where only truncating variants cause disease'; TP53 is sensitive to missense. Therefore, BP1 is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, the rule for BP2 is: 'Observed in trans with a pathogenic variant in a dominant disorder'; no such data. Therefore, BP2 is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule for BP3 is: 'In-frame indels in repetitive region'; this is a missense substitution. Therefore, BP3 is not applied.
BP4 (Not Applied)
According to VCEP guidelines, the rule for BP4 Supporting is: 'BayesDel <0.16 AND no splicing impact'; no BayesDel or splicing data are provided. Therefore, BP4 is not applied.
BP5 (Not Applied)
According to standard ACMG guidelines, the rule for BP5 is: 'Variant found in a case with an alternate molecular basis for disease'; no alternate etiology data. Therefore, BP5 is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule for BP6 is: 'Reputable source reports benign without evidence'; no benign reports. Therefore, BP6 is not applied.
BP7 (Not Applied)
According to VCEP guidelines, the rule for BP7 Supporting is: 'Synonymous or intronic change with no splicing impact'; this variant is missense. Therefore, BP7 is not applied.