Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000051.3 | RefSeq Select | 13147 nt | 386–9556 |
| NM_000051.4 | MANE Select | 12915 nt | 151–9321 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenVariant summary: ATM c.4049C>T (p.Thr1350Met) results in a non-conservative amino acid change in the encoded protein sequence. Three of five in-silico tools predict a damaging effect of the variant on protein function. The variant allele was found at a frequency of 5.2e-05 in 271256 control chromosomes (gnomAD). This frequency is not significantly higher than estimated for a pathogenic variant in ATM causing Ataxia-Telangiectasia (5.2e-05 vs 0.004), allowing no conclusion about variant significance. c.4049C>T has been reported in the literature in at least one individual affected with familial cutaneous melanoma (e.g. Pastorino_2020), and in individuals with breast cancer as well as healthy control subjects (Momozawa_2018, Kwong_2020, Dorling_2021). These reports do not provide unequivocal conclusions about association of the variant with Ataxia-Telangiectasia. To our knowledge, no experimental evidence demonstrating an impact on protein function has been reported. The following publications have been ascertained in the context of this evaluation (PMID: 30287823, 30181556, 32325837, 28119368, 33471991, 32068069). Seven submitters have cited clinical-significance assessments for this variant to ClinVar after 2014. All submitters classified the variant as uncertain significance. Based on the evidence outlined above, the variant was classified as uncertain significance.
This missense variant replaces threonine with methionine at codon 1350 of the ATM protein. Computational prediction suggests that this variant may not impact protein structure and function (internally defined REVEL score threshold <= 0.5, PMID: 27666373). To our knowledge, functional studies have not been reported for this variant. This variant has been reported in an individual affected with cutaneous melanoma (PMID: 32325837). In a large international case-control study, this variant was reported in 3/60466 breast cancer cases and 2/53461 controls (PMID: 33471991). This variant has also been identified in 13/248774 chromosomes in the general population by the Genome Aggregation Database (gnomAD). The available evidence is insufficient to determine the role of this variant in disease conclusively. Therefore, this variant is classified as a Variant of Uncertain Significance.
The p.T1350M variant (also known as c.4049C>T), located in coding exon 26 of the ATM gene, results from a C to T substitution at nucleotide position 4049. The threonine at codon 1350 is replaced by methionine, an amino acid with similar properties. This alteration was observed with an allele frequency of 0 in 7,051 unselected female breast cancer patients and was observed with an allele frequency of 0.00009 in 11,241 female controls of Japanese ancestry (Momozawa Y et al. Nat Commun, 2018 10;9:4083). This variant was also reported in 3/60,466 breast cancer cases and in 2/53,461 controls (Dorling et al. N Engl J Med. 2021 02;384:428-439). This amino acid position is highly conserved in available vertebrate species. In addition, this alteration is predicted to be deleterious by in silico analysis. Based on the available evidence, the clinical significance of this variant remains unclear.
"This variant has been reported in ClinVar as Uncertain significance (11 clinical laboratories)."
COSMIC Somatic Evidence
Open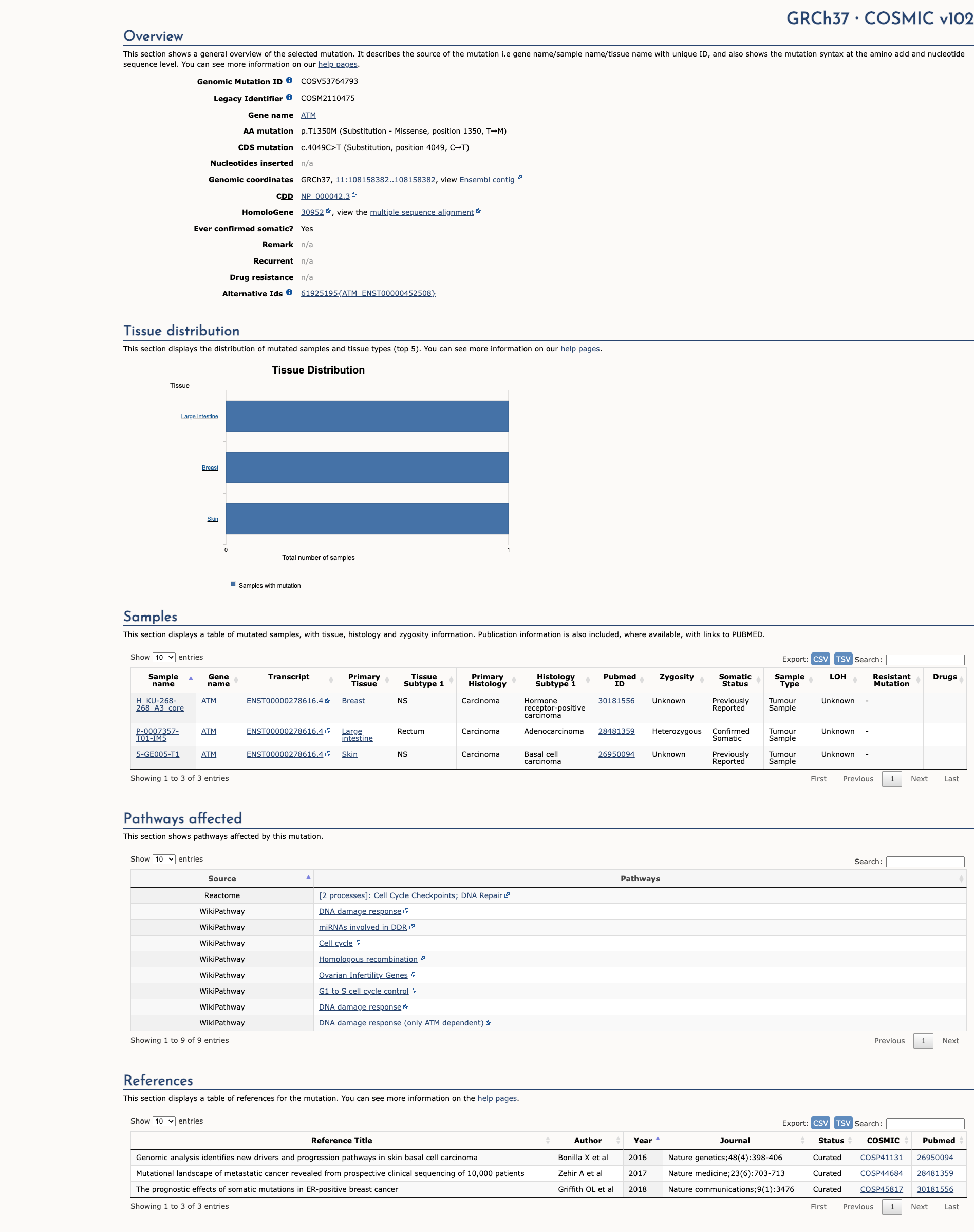
Functional Impact & Domains
Functional Domain
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | -55 bp |
| Donor Loss (DL) | 0.0 | 60 bp |
| Acceptor Gain (AG) | 0.02 | -238 bp |
| Donor Gain (DG) | 0.03 | -134 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Not Applied)
According to VCEP guidelines, PVS1 is applied only to null variants as per the ATM PVS1 Decision Tree. The evidence shows a missense variant (p.T1350M). Therefore, PVS1 is not applied.
PS1 (Not Applied)
According to VCEP guidelines, PS1 requires the same amino acid change as a previously established pathogenic variant. There is no known pathogenic variant with the same amino acid change. Therefore, PS1 is not applied.
PS2 (Not Applied)
According to standard ACMG guidelines, PS2 requires confirmed de novo occurrence. There is no de novo evidence. Therefore, PS2 is not applied.
PS3 (Not Applied)
According to VCEP guidelines, PS3 requires functional studies showing failure to rescue ATM-specific features. No functional characterization is available. Therefore, PS3 is not applied.
PS4 (Not Applied)
According to VCEP guidelines, PS4 requires case-control data with significant association. No case-control or proband data are available. Therefore, PS4 is not applied.
PM1 (Not Applied)
According to standard ACMG guidelines, PM1 requires localization in a mutational hot spot or functional domain. There is no evidence that p.T1350M falls in such a region. Therefore, PM1 is not applied.
PM2 (Not Applied)
According to VCEP guidelines, PM2_Supporting applies only for frequency ≤0.001% with n=1. The variant MAF=0.00523% with multiple observations. Therefore, PM2 is not applied.
PM3 (Not Applied)
According to VCEP guidelines, PM3 applies to variants detected in trans with a pathogenic variant in recessive disease. No trans data are available. Therefore, PM3 is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, PM4 applies to protein length changes such as in-frame indels or stop-loss. The variant is a missense change. Therefore, PM4 is not applied.
PM5 (Not Applied)
According to VCEP guidelines, PM5_Supporting applies to novel missense at the same codon as a known pathogenic truncating variant upstream of p.R3047. There is no such upstream truncating pathogenic variant at codon 1350. Therefore, PM5 is not applied.
PM6 (Not Applied)
According to standard ACMG guidelines, PM6 requires assumed de novo occurrence without confirmation. No de novo data are available. Therefore, PM6 is not applied.
PP1 (Not Applied)
According to standard ACMG guidelines, PP1 requires segregation data. No segregation information is available. Therefore, PP1 is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, PP2 applies to missense variants in genes with low benign variation and where missense is a common mechanism. ATM has known pathogenic missense and benign variation patterns are unclear. Therefore, PP2 is not applied.
PP3 (Not Applied)
According to VCEP guidelines, PP3_Supporting requires REVEL >0.7333 or RNA impact. The REVEL score is 0.39 and SpliceAI predicts no splicing impact. Therefore, PP3 is not applied.
PP4 (Not Applied)
According to standard ACMG guidelines, PP4 requires a patient phenotype highly specific for a disease. No phenotype data are provided. Therefore, PP4 is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, PP5 requires a reputable source classifying as pathogenic. ClinVar reports this variant as VUS. Therefore, PP5 is not applied.
BA1 (Not Applied)
According to VCEP guidelines, BA1 requires allele frequency >5%. The MAF is 0.00523%. Therefore, BA1 is not applied.
BS1 (Not Applied)
According to VCEP guidelines, BS1 requires allele frequency >0.05%. The MAF is 0.00523%. Therefore, BS1 is not applied.
BS2 (Not Applied)
According to standard ACMG guidelines, BS2 requires observation in healthy adults inconsistent with disease. No such data are available. Therefore, BS2 is not applied.
BS3 (Not Applied)
According to VCEP guidelines, BS3 requires functional rescue of ATM-specific features. No functional studies show rescue. Therefore, BS3 is not applied.
BS4 (Not Applied)
According to standard ACMG guidelines, BS4 requires lack of segregation in affected individuals. No segregation data are available. Therefore, BS4 is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, BP1 applies to missense in genes where only truncating variants cause disease. ATM has known pathogenic missense variants. Therefore, BP1 is not applied.
BP2 (Not Applied)
According to VCEP guidelines, BP2 applies to variants in trans with a pathogenic variant in recessive disease. No trans data are available. Therefore, BP2 is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, BP3 applies to in-frame indels in repetitive regions. This is a missense change. Therefore, BP3 is not applied.
BP4 (Supporting)
According to standard ACMG guidelines, BP4 is supported by multiple computational lines of evidence suggesting no impact. In silico tools (CADD, MetaSVM, MetaLR, PrimateAI) predict benign impact and SpliceAI predicts no splicing effect. Therefore, BP4 is applied at Supporting strength.
BP5 (Not Applied)
According to standard ACMG guidelines, BP5 applies when a variant is found in cis with a pathogenic variant in a patient with an alternate molecular cause. No such data are available. Therefore, BP5 is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, BP6 requires a reputable source reporting as benign. No such classification exists. Therefore, BP6 is not applied.
BP7 (Not Applied)
According to standard ACMG guidelines, BP7 applies to synonymous or deep intronic variants with no splicing impact. This is a missense change. Therefore, BP7 is not applied.