Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000059.4 | MANE Select | 11954 nt | 200–10456 |
| NM_000059.2 | Alternative | 11386 nt | 228–10484 |
| NM_000059.3 | RefSeq Select | 11386 nt | 228–10484 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThis variant deletes 2 nucleotides in exon 11 of the BRCA2 gene, creating a frameshift and premature translation stop signal. This variant is expected to result in an absent or non-functional protein product. This variant has been reported in individuals affected with breast and/or ovarian cancer and in high-risk breast and ovarian cancer families (PMID: 19656164, 22217648, 22382806, 22798144, 25863477, 26187060, 27836010, doi:10.21203/rs.3.rs-734551/v1), and has also been identified in 6 families among the CIMBA participants (PMID: 29446198) (https://cimba.ccge.medschl.cam.ac.uk/). This variant has been identified in 1/251336 chromosomes in the general population by the Genome Aggregation Database (gnomAD). Loss of BRCA2 function is a known mechanism of disease (clinicalgenome.org). Based on the available evidence, this variant is classified as Pathogenic.
This sequence change creates a premature translational stop signal (p.Asp2242Phefs*2) in the BRCA2 gene. It is expected to result in an absent or disrupted protein product. Loss-of-function variants in BRCA2 are known to be pathogenic (PMID: 20104584). This variant is present in population databases (rs397507375, gnomAD 0.003%). This premature translational stop signal has been observed in individual(s) with breast cancer (PMID: 19656164, 22217648, 22382806, 22798144, 25863477). This variant is also known as 6952delGA. ClinVar contains an entry for this variant (Variation ID: 38062). For these reasons, this variant has been classified as Pathogenic.
The c.6724_6725delGA pathogenic mutation, located in coding exon 10 of the BRCA2 gene, results from a deletion of two nucleotides at nucleotide positions 6724 to 6725, causing a translational frameshift with a predicted alternate stop codon (p.D2242Ffs*2). This variant was reported in individual(s) with features consistent with BRCA2-related cancer predisposition (Kwong A et al. J Med Genet. 2016 Jan;53:15-23; Rebbeck TR et al. Hum. Mutat. 2018 May;39:593-620; Ryu JM et al. Breast Cancer Res Treat. 2019 Jan;173:385-395). In addition to the clinical data presented in the literature, this alteration is expected to result in loss of function by premature protein truncation or nonsense-mediated mRNA decay. As such, this alteration is interpreted as a disease-causing mutation.
This variant alters the translational reading frame of the BRCA2 mRNA and causes the premature termination of BRCA2 protein synthesis. Additionally, this variant has been reported in an individual with breast cancer in the published literature (PMID: 19656164 (2009)). Based on the available information, this variant is classified as pathogenic.
Variant summary: BRCA2 c.6724_6725delGA (p.Asp2242PhefsX2) results in a premature termination codon, predicted to cause a truncation of the encoded protein or absence of the protein due to nonsense mediated decay, which are commonly known mechanisms for disease. Variants downstream of this position have been classified as pathogenic by our laboratory. The variant allele was found at a frequency of 4e-06 in 251336 control chromosomes. The available data on variant occurrences in the general population are insufficient to allow any conclusion about variant significance. c.6724_6725delGA has been reported in the literature in multiple individuals affected with Hereditary Breast And Ovarian Cancer Syndrome (e.g., Kang_2015). These data indicate that the variant is very likely to be associated with disease. To our knowledge, no experimental evidence demonstrating an impact on protein function has been reported. The following publication was ascertained in the context of this evaluation (PMID: 25863477). Seven ClinVar submitters (evaluation after 2014) have cited the variant, and all submitters classified the variant as pathogenic. Based on the evidence outlined above, the variant was classified as pathogenic.
"This variant has been reported in ClinVar as Pathogenic (12 clinical laboratories) and as Pathogenic by Evidence-based Network for the Interpretation of Germline Mutant Alleles (ENIGMA) expert panel."
COSMIC Somatic Evidence
Open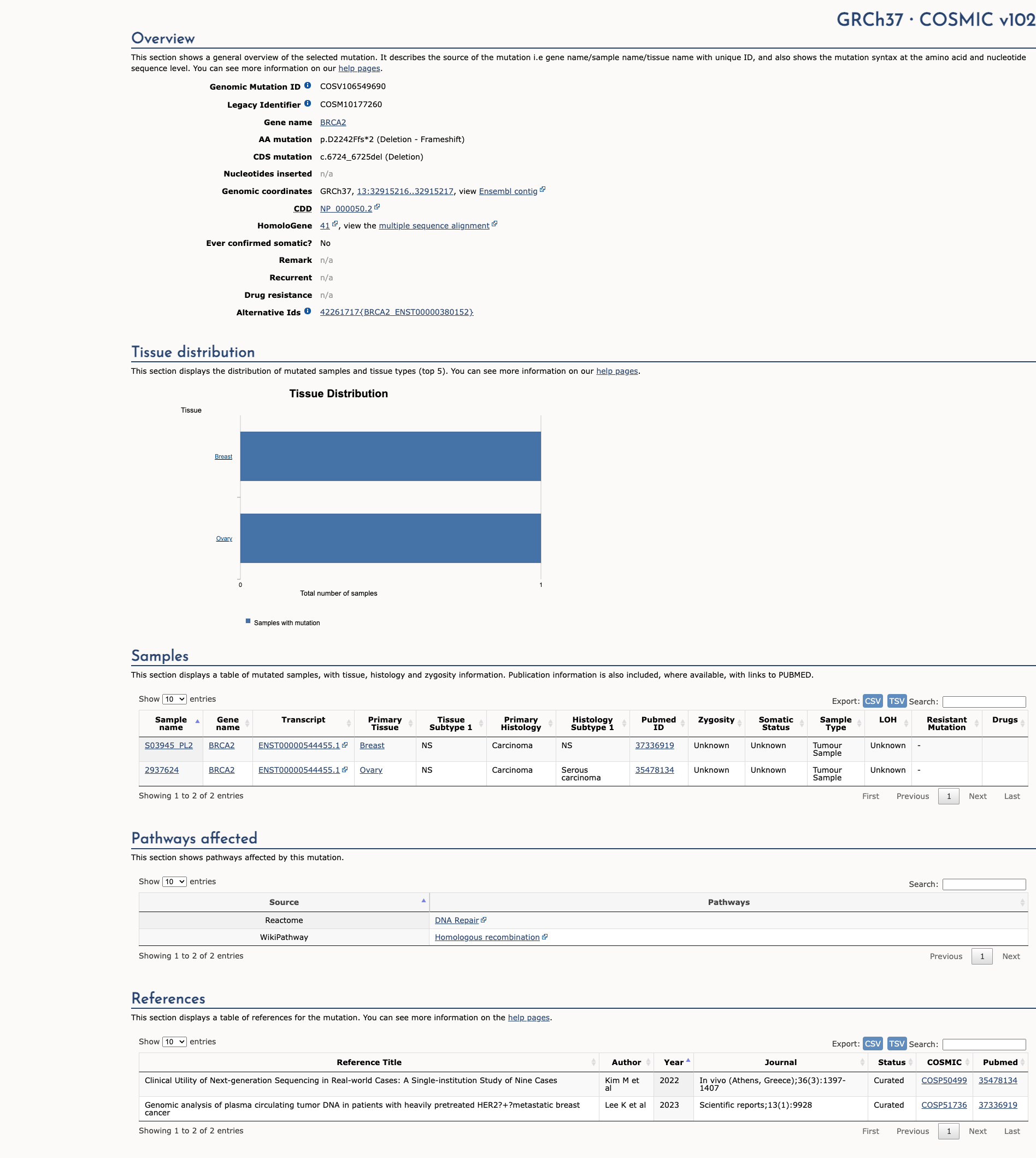
Functional Impact & Domains
Functional Domain
The BRCA2 D2242Ffs*2 variant is a truncating mutation that likely results in loss of function. Functional studies indicate that truncating mutations in BRCA2 impair nuclear localization, which is essential for its role in the DNA damage response. This disruption affects the integrity of homologous recombination, a critical process for DNA repair. Consequently, such mutations are associated with an increased risk of developing various cancers, including breast, ovarian, prostate, and pancreatic cancers.
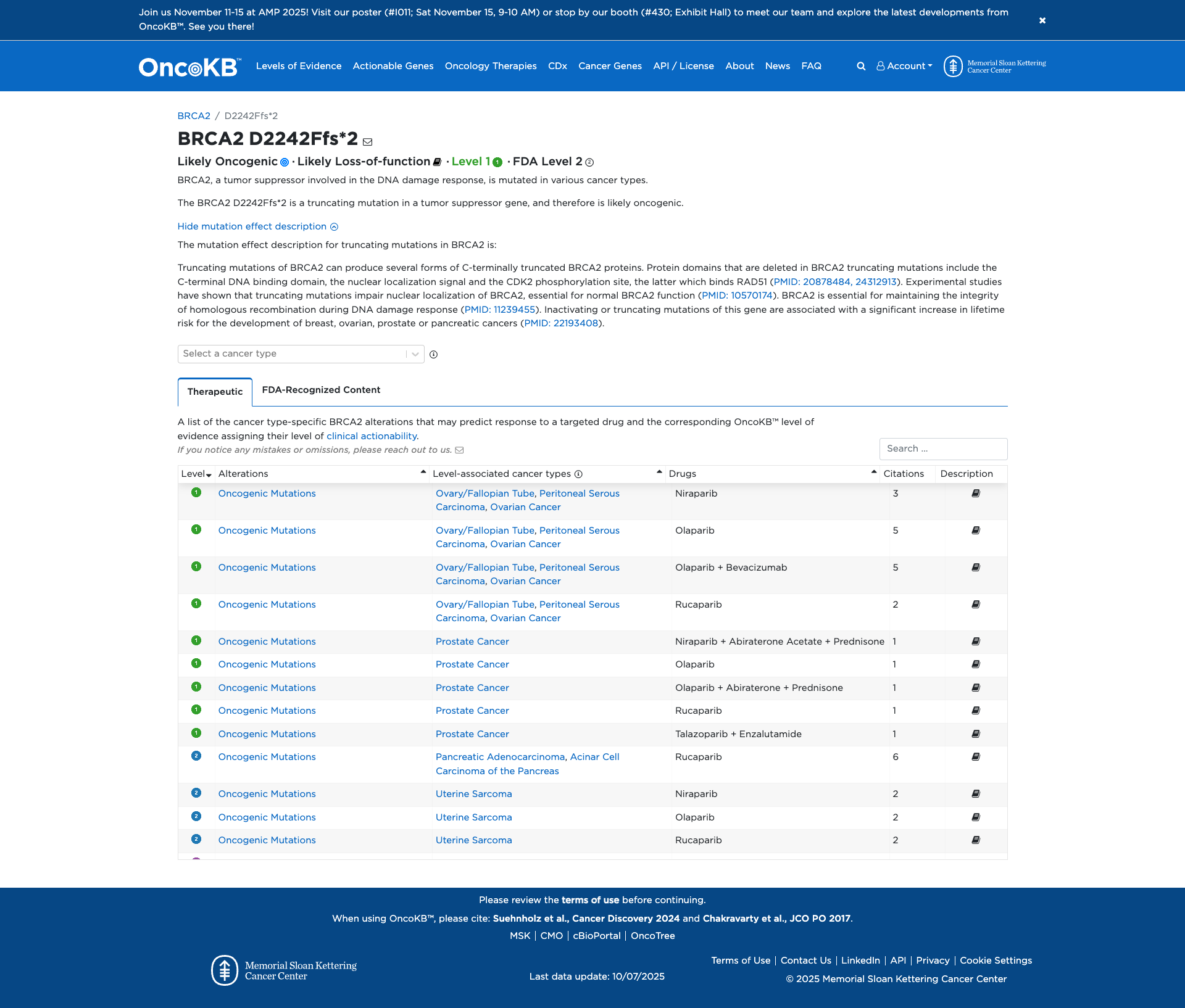

Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.03 | -69 bp |
| Donor Loss (DL) | 0.02 | 119 bp |
| Acceptor Gain (AG) | 0.0 | 59 bp |
| Donor Gain (DG) | 0.0 | 115 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Very Strong)
According to VCEP guidelines, the rule/finding for PVS1 is: "**Very Strong Strength**: Very Strong Null variant (nonsense, frameshift, splice site (donor/acceptor +/−1,2), initiation codon, single or multi-exon deletion) in a gene where loss of function (LOF) is a known mechanism of disease. Apply at appropriate strength according to PVS1 flowchart...". The evidence for this variant shows: c.6724_6725del (D2242Ffs*2) is a frameshift predicted to create a premature termination codon in BRCA2, a gene where LOF is a known mechanism of disease and the variant is not in the last exon. Therefore, this criterion is applied at Very Strong strength because it is a null variant in a gene where LOF causes disease.
PS1 (Not Applied)
According to VCEP guidelines, the rule/finding for PS1 is: "**Strong Strength**: Strong Apply PS1 for predicted missense substitutions where a previously classified pathogenic variant is considered to act via protein change (no confirmed or predicted effect on mRNA splicing (SpliceAI≤0.1))." The evidence for this variant shows: c.6724_6725del is a frameshift variant, not a missense substitution. Therefore, this criterion is not applied.
PS2 (Not Applied)
According to standard ACMG guidelines, the rule/finding for PS2 is: "De novo (both maternity and paternity confirmed) in a patient with the disease and no family history." The evidence for this variant shows: no de novo information is available. Therefore, this criterion is not applied.
PS3 (Strong)
According to VCEP guidelines, the rule/finding for PS3 is: "**Strong Strength**: Strong Well-established in vitro or in vivo functional studies supportive of a damaging effect. Apply PS3 for assays measuring effect via protein only OR mRNA and protein combined." The evidence for this variant shows: functional studies indicate truncating BRCA2 mutations impair nuclear localization and homologous recombination. Therefore, this criterion is applied at Strong strength because well-established functional data support a damaging effect.
PS4 (Not Applied)
According to VCEP guidelines, the rule/finding for PS4 is: "**Strong Strength**: Strong The prevalence of the variant in affected individuals is significantly increased compared to the prevalence in controls. Case-control studies; p-value ≤0.05 and OR ≥4 (lower confidence interval excludes 2.0)." The evidence for this variant shows: no case-control data are available. Therefore, this criterion is not applied.
PM1 (Not Applied)
According to VCEP guidelines, PM1 applies to: "Variants located in a (potentially) clinically important functional domain defined for BRCA2." The evidence for this variant shows: it is a truncating variant, not evaluated by domain-based criteria. Therefore, this criterion is not applied.
PM2 (Supporting)
According to VCEP guidelines, the rule/finding for PM2 is: "**Supporting Strength**: Supporting Absent from controls in an outbred population, from gnomAD v2.1 (non-cancer, exome only subset) and gnomAD v3.1 (non-cancer)." The evidence for this variant shows: MAF = 0.000398% in gnomAD, extremely rare. Therefore, this criterion is applied at Supporting strength because the variant is absent or at extremely low frequency in controls.
PM3 (Not Applied)
According to VCEP guidelines, PM3 applies to: "Variants observed in trans with a pathogenic variant in BRCA1/2-related Fanconi Anemia phenotype with phenotype-specific points." The evidence for this variant shows: no biallelic or FA phenotype data available. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, the rule/finding for PM4 is: "Protein length changes as a result of in-frame deletions/insertions in a non-repeat region or stop-loss variants." The evidence for this variant shows: this is a frameshift/truncating variant covered under PVS1. Therefore, this criterion is not applied.
PM5 (Not Applied)
According to VCEP guidelines, the rule/finding for PM5 is: "**Strong Strength**: Strong Protein termination codon (PTC) variant in an exon where a different proven pathogenic PTC variant has been seen before." The evidence for this variant shows: no data are available demonstrating another PTC in the same exon. Therefore, this criterion is not applied.
PM6 (Not Applied)
According to standard ACMG guidelines, the rule/finding for PM6 is: "Assumed de novo, but without confirmation of paternity and maternity." The evidence for this variant shows: no de novo information is available. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to VCEP guidelines, the rule/finding for PP1 is: "Co-segregation with disease in multiple affected family members in BRCA2, as measured by a quantitative co-segregation analysis." The evidence for this variant shows: no segregation data are available. Therefore, this criterion is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, the rule/finding for PP2 is: "Missense variant in a gene with a low rate of benign missense variation and where missense variants are a common mechanism of disease." The evidence for this variant shows: this is a frameshift variant, not missense. Therefore, this criterion is not applied.
PP3 (Not Applied)
According to VCEP guidelines, the rule/finding for PP3 is: "Supporting predicted splicing impact (SpliceAI ≥0.2) or protein impact (BayesDel no-AF ≥0.30) for missense/in-frame variants in functional domains." The evidence for this variant shows: frameshift variants are not evaluated by in silico predictors. Therefore, this criterion is not applied.
PP4 (Not Applied)
According to VCEP guidelines, the rule/finding for PP4 is: "Use of specific patient phenotype/family history likelihood ratios for BRCA2-related cancer phenotypes." The evidence for this variant shows: individual clinical phenotype data are not provided. Therefore, this criterion is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, the rule/finding for PP5 is: "Reputable source reports variant as pathogenic, but evidence is not available for independent evaluation." The evidence for this variant shows: although ClinVar and ENIGMA report pathogenicity, PP5 is discouraged by VCEP. Therefore, this criterion is not applied.
BA1 (Not Applied)
According to VCEP guidelines, the rule/finding for BA1 is: "Stand Alone filter allele frequency (FAF) >0.001 in gnomAD non-cancer populations." The evidence for this variant shows: MAF = 0.000398%, below threshold. Therefore, this criterion is not applied.
BS1 (Not Applied)
According to VCEP guidelines, the rule/finding for BS1 is: "Strong FAF >0.0001 and ≤0.001; Supporting FAF >0.00002 and ≤0.0001 in gnomAD non-cancer populations." The evidence for this variant shows: MAF = 0.000398%, below strong threshold and above supporting but not used for BS1. Therefore, this criterion is not applied.
BS2 (Not Applied)
According to VCEP guidelines, the rule/finding for BS2 is: "Observed in healthy individuals without Fanconi Anemia features, with points-based system." The evidence for this variant shows: no data on FA phenotype in healthy adults. Therefore, this criterion is not applied.
BS3 (Not Applied)
According to VCEP guidelines, the rule/finding for BS3 is: "Strong Well-established functional studies show no damaging effect on protein function." The evidence for this variant shows: functional studies demonstrate damaging effect. Therefore, this criterion is not applied.
BS4 (Not Applied)
According to VCEP guidelines, the rule/finding for BS4 is: "Strong Lack of segregation in affected members as measured by Bayes Score ≤0.05." The evidence for this variant shows: no segregation data available. Therefore, this criterion is not applied.
BP1 (Not Applied)
According to VCEP guidelines, the rule/finding for BP1 is: "Strong for silent or missense variants outside functional domains without splicing impact." The evidence for this variant shows: it is a truncating variant, not silent or missense. Therefore, this criterion is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, the rule/finding for BP2 is: "Observed in trans with a pathogenic variant for a fully penetrant autosomal dominant gene." The evidence for this variant shows: no data on co-occurrence. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule/finding for BP3 is: "In-frame indels in repetitive region without a known function." The evidence for this variant shows: it is a frameshift variant, not an in-frame indel. Therefore, this criterion is not applied.
BP4 (Not Applied)
According to VCEP guidelines, the rule/finding for BP4 is: "Supporting no predicted impact via protein or splicing for missense/in-frame variants inside functional domains." The evidence for this variant shows: it is a frameshift, not suitable for computational benign prediction. Therefore, this criterion is not applied.
BP5 (Not Applied)
According to VCEP guidelines, the rule/finding for BP5 is: "Use to capture LR against pathogenicity based on multifactorial data for co-observed variants in different cancer genes." The evidence for this variant shows: no such co-observation data. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule/finding for BP6 is: "Reputable source reports variant as benign, but evidence is not available." The evidence for this variant shows: no reputable benign report. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to VCEP guidelines, the rule/finding for BP7 is: "Supporting for silent or intronic variants outside splice sites with no predicted impact." The evidence for this variant shows: it is a frameshift variant, not silent or intronic. Therefore, this criterion is not applied.