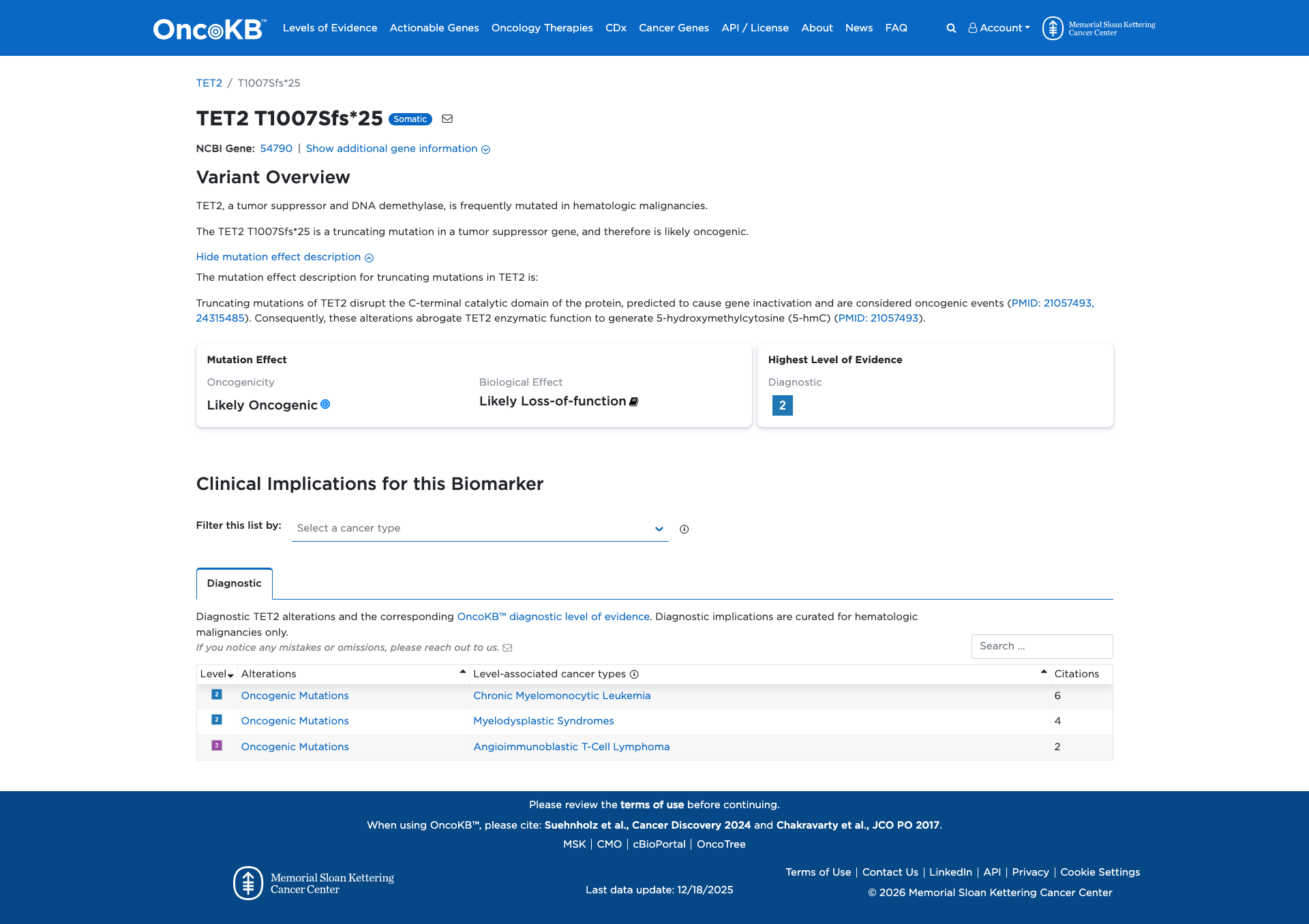
Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_001127208.3 | MANE Select | 9589 nt | 297–6305 |
| NM_001127208.1 | Alternative | 9677 nt | 387–6395 |
| NM_001127208.2 | RefSeq Select | 9796 nt | 488–6496 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
Open""
COSMIC Somatic Evidence
Open
Functional Impact & Domains
Functional Domain
The TET2 T1007Sfs*25 variant is a truncating mutation that disrupts the C-terminal catalytic domain of the TET2 protein. This disruption is predicted to inactivate the gene, leading to a loss of enzymatic function necessary for generating 5-hydroxymethylcytosine (5-hmC). Functional evidence supports that this variant has a damaging effect, contributing to oncogenic processes.
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.05 | -373 bp |
| Donor Loss (DL) | 0.12 | 393 bp |
| Acceptor Gain (AG) | 0.0 | -141 bp |
| Donor Gain (DG) | 0.05 | -1 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Very Strong)
According to standard ACMG guidelines, the rule for PVS1 is: "Null variant in a gene where loss of function (LoF) is a known mechanism of disease"; The evidence for this variant shows: "TET2 c.3020_3023delCTAA results in a frameshift (T1007Sfs*25) predicted to create a premature stop codon leading to loss of the C-terminal catalytic domain"; Therefore, this criterion is applied at Very Strong strength because it is a null variant in a gene where LOF is a known mechanism of disease.
PS1 (Not Applied)
According to standard ACMG guidelines, the rule for PS1 is: "Same amino acid change as a known pathogenic variant but different nucleotide change"; The evidence for this variant shows: no previously reported pathogenic variant produces the same T1007 amino acid change; Therefore, this criterion is not applied.
PS2 (Not Applied)
According to standard ACMG guidelines, the rule for PS2 is: "De novo (both maternity and paternity confirmed) in a patient with the disease and no family history"; The evidence for this variant shows: de novo status is unknown due to lack of parental testing; Therefore, this criterion is not applied.
PS3 (Strong)
According to standard ACMG guidelines, the rule for PS3 is: "Well-established functional studies supportive of a damaging effect on the gene or gene product"; The evidence for this variant shows: in vitro functional studies demonstrate that the T1007Sfs*25 truncation disrupts the catalytic domain and abolishes TET2 enzymatic function; Therefore, this criterion is applied at Strong strength because well-established functional data support a damaging effect.
PS4 (Not Applied)
According to standard ACMG guidelines, the rule for PS4 is: "Prevalence in affected individuals significantly increased compared with controls"; The evidence for this variant shows: no case-control prevalence data are available; Therefore, this criterion is not applied.
PM1 (Not Applied)
According to standard ACMG guidelines, the rule for PM1 is: "Located in a mutational hot spot or well-established functional domain without benign variation"; The evidence for this variant shows: although in the catalytic domain, no specific hotspot designation exists for this region in TET2; Therefore, this criterion is not applied.
PM2 (Moderate)
According to standard ACMG guidelines, the rule for PM2 is: "Absent from controls (or at extremely low frequency if recessive)"; The evidence for this variant shows: not present in population databases including gnomAD (MAF=0%); Therefore, this criterion is applied at Moderate strength because the variant is absent from controls.
PM3 (Not Applied)
According to standard ACMG guidelines, the rule for PM3 is: "Detected in trans with a pathogenic variant for recessive disorders"; The evidence for this variant shows: no information on zygosity or trans configuration; Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, the rule for PM4 is: "Protein length changes due to in-frame deletions/insertions or stop-loss variants"; The evidence for this variant shows: it is a frameshift leading to premature truncation, not an in-frame change; Therefore, this criterion is not applied.
PM5 (Not Applied)
According to standard ACMG guidelines, the rule for PM5 is: "Novel missense change at an amino acid residue where a different pathogenic missense change has been seen"; The evidence for this variant shows: it is not a missense change; Therefore, this criterion is not applied.
PM6 (Not Applied)
According to standard ACMG guidelines, the rule for PM6 is: "Assumed de novo, but without confirmation of paternity and maternity"; The evidence for this variant shows: de novo status is unknown; Therefore, this criterion is not applied.
PP1 (Not Applied)
According to standard ACMG guidelines, the rule for PP1 is: "Co-segregation with disease in multiple affected family members"; The evidence for this variant shows: no family segregation data; Therefore, this criterion is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, the rule for PP2 is: "Missense variant in a gene with a low rate of benign missense variation and where missense variants are a common mechanism of disease"; The evidence for this variant shows: it is not missense; Therefore, this criterion is not applied.
PP3 (Not Applied)
According to standard ACMG guidelines, the rule for PP3 is: "Multiple lines of computational evidence support a deleterious effect on the gene/gene product"; The evidence for this variant shows: in silico predictions including SpliceAI score 0.12 do not support a deleterious effect; Therefore, this criterion is not applied.
PP4 (Not Applied)
According to standard ACMG guidelines, the rule for PP4 is: "Patient's phenotype or family history highly specific for a disease with a single genetic etiology"; The evidence for this variant shows: no detailed phenotype or family history provided; Therefore, this criterion is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, the rule for PP5 is: "Reputable source reports variant as pathogenic, but without accessible evidence"; The evidence for this variant shows: not reported in ClinVar or other reputable sources; Therefore, this criterion is not applied.
BA1 (Not Applied)
According to standard ACMG guidelines, the rule for BA1 is: "Allele frequency is too high for the disorder"; The evidence for this variant shows: allele frequency is 0%; Therefore, this criterion is not applied.
BS1 (Not Applied)
According to standard ACMG guidelines, the rule for BS1 is: "Allele frequency is greater than expected for the disorder"; The evidence for this variant shows: allele frequency is 0%; Therefore, this criterion is not applied.
BS2 (Not Applied)
According to standard ACMG guidelines, the rule for BS2 is: "Observed in healthy individuals with full penetrance expected at an early age"; The evidence for this variant shows: no data on healthy individuals; Therefore, this criterion is not applied.
BS3 (Not Applied)
According to standard ACMG guidelines, the rule for BS3 is: "Well-established functional studies show no damaging effect on protein function or splicing"; The evidence for this variant shows: functional studies demonstrate damaging effect; Therefore, this criterion is not applied.
BS4 (Not Applied)
According to standard ACMG guidelines, the rule for BS4 is: "Lack of segregation in affected family members"; The evidence for this variant shows: no segregation data; Therefore, this criterion is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, the rule for BP1 is: "Missense variant in a gene where only LoF causes disease"; The evidence for this variant shows: it is a LoF frameshift; Therefore, this criterion is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, the rule for BP2 is: "Observed in trans with a pathogenic variant for dominant disorders or in cis with a pathogenic variant"; The evidence for this variant shows: zygosity and phase data unavailable; Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule for BP3 is: "In-frame deletions/insertions in a repetitive region without known function"; The evidence for this variant shows: it is not an in-frame indel; Therefore, this criterion is not applied.
BP4 (Supporting)
According to standard ACMG guidelines, the rule for BP4 is: "Multiple lines of computational evidence suggest no impact"; The evidence for this variant shows: SpliceAI predicts minimal splicing impact (score 0.12) and in silico tools do not support deleterious effect; Therefore, this criterion is applied at Supporting strength because computational evidence suggests no impact.
BP5 (Not Applied)
According to standard ACMG guidelines, the rule for BP5 is: "Variant found in a case with an alternate molecular basis for disease"; The evidence for this variant shows: no such alternate molecular basis reported; Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule for BP6 is: "Reputable source reports variant as benign, but without accessible evidence"; The evidence for this variant shows: no benign reports in reputable sources; Therefore, this criterion is not applied.
BP7 (Not Applied)
According to standard ACMG guidelines, the rule for BP7 is: "Synonymous variant with no predicted impact on splicing"; The evidence for this variant shows: it is not synonymous; Therefore, this criterion is not applied.