Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000314.8 | MANE Select | 8515 nt | 846–2057 |
| NM_000314.7 | RefSeq Select | 8514 nt | 845–2056 |
| NM_000314.5 | Alternative | 8719 nt | 1032–2243 |
| NM_000314.4 | Alternative | 5572 nt | 1032–2243 |
| NM_000314.3 | Alternative | 3416 nt | 1032–2243 |
| NM_000314.6 | Alternative | 8718 nt | 1032–2243 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThe p.S287* pathogenic mutation (also known as c.860C>G), located in coding exon 8 of the PTEN gene, results from a C to G substitution at nucleotide position 860. This changes the amino acid from a serine to a stop codon within coding exon 8. This alteration has been reported as a de novo finding in a patient with macrocephaly (Lee H et al. JAMA, 2014 Nov;312:1880-7). This variant is considered to be rare based on population cohorts in the Genome Aggregation Database (gnomAD). This alteration is expected to result in loss of function by premature protein truncation or nonsense-mediated mRNA decay. As such, this alteration is interpreted as a disease-causing mutation.
This sequence change creates a premature translational stop signal (p.Ser287*) in the PTEN gene. It is expected to result in an absent or disrupted protein product. Loss-of-function variants in PTEN are known to be pathogenic (PMID: 9467011, 21194675). This variant is not present in population databases (gnomAD no frequency). This premature translational stop signal has been observed in individual(s) with PTEN hamartoma tumor syndrome (PMID: 25326637). ClinVar contains an entry for this variant (Variation ID: 216987). For these reasons, this variant has been classified as Pathogenic.
"This variant has been reported in ClinVar as Pathogenic (3 clinical laboratories) and as Likely pathogenic (1 clinical laboratories)."
COSMIC Somatic Evidence
Open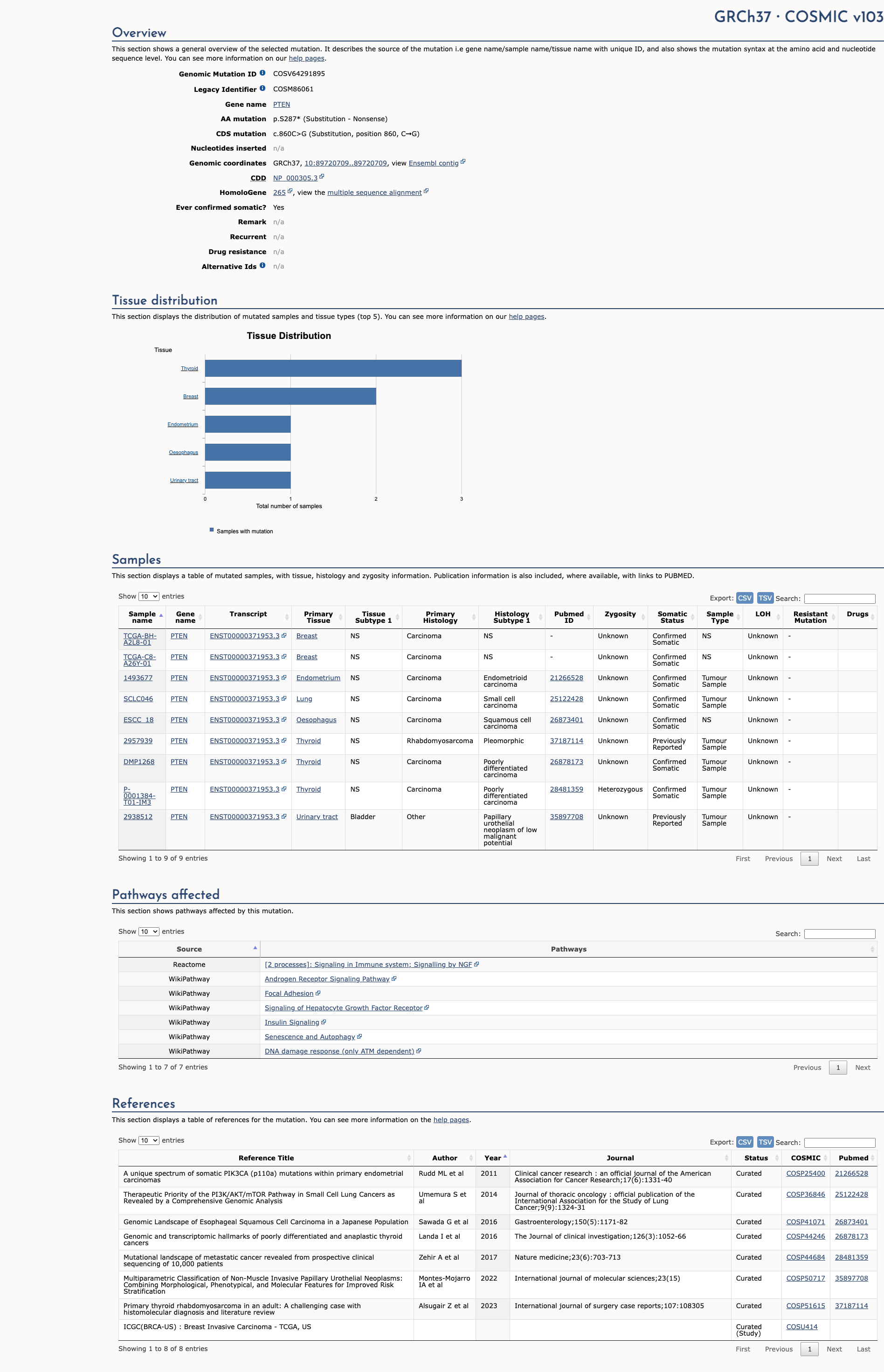
Functional Impact & Domains
Functional Domain
The PTEN S287* variant is a truncating mutation that results in the premature termination of the PTEN protein, leading to the loss of its phosphatase function. Functional studies indicate that similar truncating mutations in PTEN result in the inability to negatively regulate the PI3K/AKT pathway and increase genomic instability. Therefore, the PTEN S287* variant is functionally characterized as damaging, likely contributing to oncogenic processes.
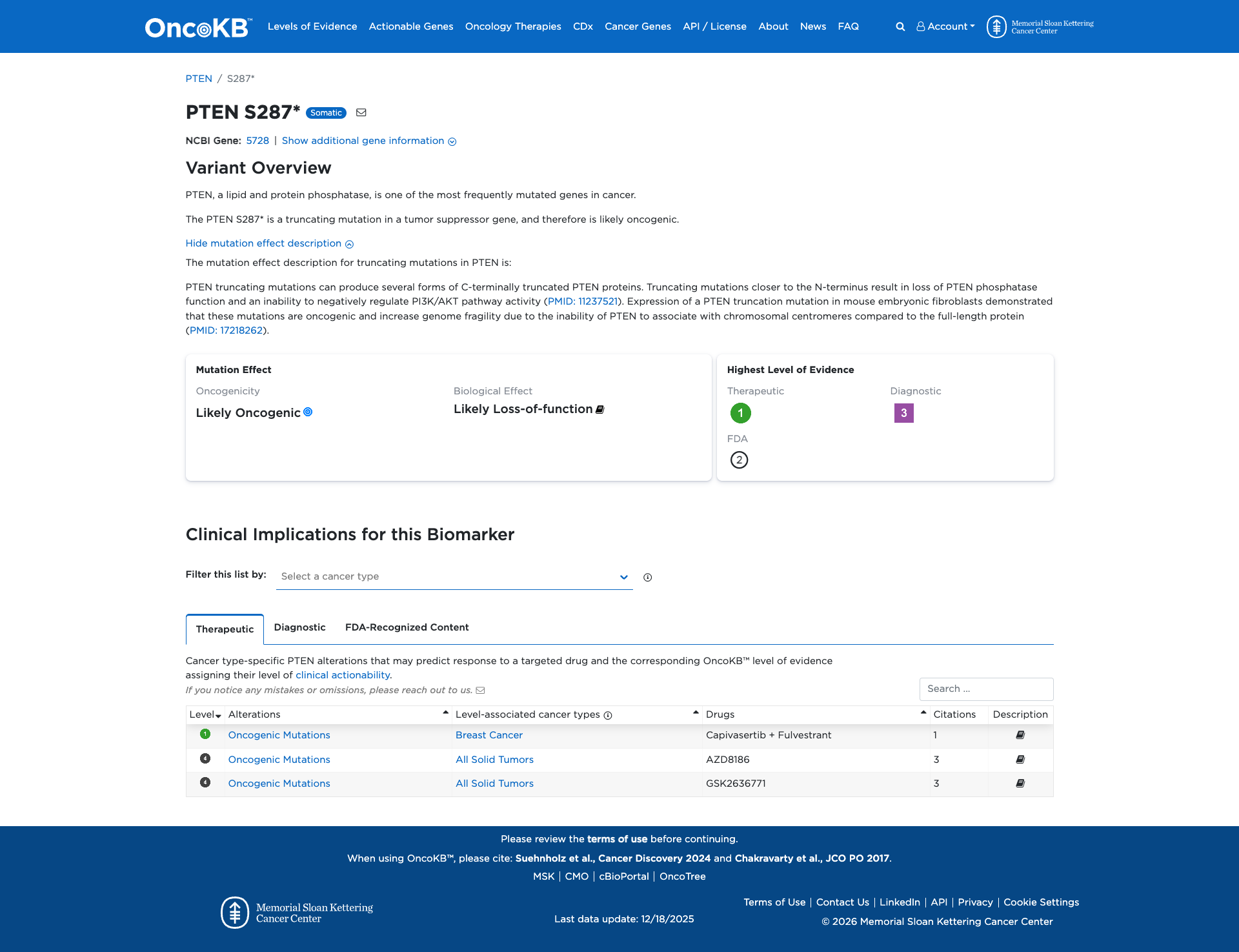
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | -15 bp |
| Donor Loss (DL) | 0.0 | -38 bp |
| Acceptor Gain (AG) | 0.0 | -9 bp |
| Donor Gain (DG) | 0.0 | 411 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Very Strong)
According to VCEP guidelines, the rule for PVS1 is: "Very Strong Strength: Very Strong Use PTEN PVS1 decision tree. Modification Type: Disease-specific". The evidence for this variant shows: c.860C>G (p.S287*) is a nonsense variant leading to loss-of-function in PTEN, where LOF is a known disease mechanism and the change is not in the last exon. Therefore, this criterion is applied at Very Strong strength because it meets the PTEN-specific PVS1 decision tree for a truncating variant in a non–last exon.
PS1 (Not Applied)
According to VCEP guidelines, the rule for PS1 is: "Strong Strength: Strong Same amino acid change as a previously established pathogenic variant regardless of nucleotide change OR different variant at same nucleotide position as a pathogenic splicing variant...". The evidence for this variant shows: no other variant produces the same amino acid change at codon 287. Therefore, this criterion is not applied.
PS2 (Not Applied)
According to VCEP guidelines, the rule for PS2 includes de novo observations required at Strong or Very Strong strength. The evidence for this variant shows: no parental or de novo data are available. Therefore, this criterion is not applied.
PS3 (Not Applied)
According to PTEN Pre-processing, the finding for PS3 is: "High confidence not TRUE ('FALSE'), PS3 rule not applied". The evidence for this variant shows: functional pre-processing did not meet the PTEN-specific high-confidence threshold. Therefore, this criterion is not applied.
PS4 (Not Applied)
According to VCEP guidelines, the rule for PS4 includes proband specificity scores or case–control enrichment. The evidence for this variant shows: no data on the number of probands or case–control studies. Therefore, this criterion is not applied.
PM1 (Not Applied)
According to VCEP guidelines, the rule for PM1 is: "Moderate Strength: Located in a mutational hot spot and/or critical functional domain defined to include residues...". The evidence for this variant shows: codon 287 is outside the defined catalytic motifs. Therefore, this criterion is not applied.
PM2 (Supporting)
According to VCEP guidelines, the rule for PM2 is: "Supporting Strength: Absent in population Databases present at <0.00001 (0.001%) allele frequency in gnomAD...". The evidence for this variant shows: not found in gnomAD or other large population databases. Therefore, this criterion is applied at Supporting strength.
PM3 (Not Applied)
According to VCEP guidelines, the rule for PM3 applies to recessive disorders requiring trans observations. The evidence for this variant shows: no evidence of recessive inheritance or trans configuration. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to VCEP guidelines, the rule for PM4 is: "Moderate Strength: Protein length changes due to in-frame indels or stop-loss variants...". The evidence for this variant shows: this is a nonsense variant, not an in-frame or stop-loss change. Therefore, this criterion is not applied.
PM5 (Not Applied)
According to VCEP guidelines, the rule for PM5 is: "Moderate Strength: Missense change at a residue where another missense change is pathogenic...". The evidence for this variant shows: this is not a missense variant. Therefore, this criterion is not applied.
PM6 (Not Applied)
According to VCEP guidelines, the rule for PM6 covers presumed de novo occurrences. The evidence for this variant shows: no de novo data. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to VCEP guidelines, the rule for PP1 requires co-segregation data. The evidence for this variant shows: no family segregation information. Therefore, this criterion is not applied.
PP2 (Not Applied)
According to VCEP guidelines, the rule for PP2 is: "Supporting Strength: Missense variant in gene with low benign missense variation...". The evidence for this variant shows: this is a truncating variant, not missense. Therefore, this criterion is not applied.
PP3 (Not Applied)
According to VCEP guidelines, the rule for PP3 is: "Supporting Strength: Multiple lines of computational evidence support a deleterious effect...". The evidence for this variant shows: in silico scores (CADD 9.65, SpliceAI 0) do not predict a deleterious effect beyond the truncation itself. Therefore, this criterion is not applied.
PP4 (Not Applied)
According to VCEP guidelines, the rule for PP4 requires a highly specific phenotype. The evidence for this variant shows: no phenotypic details provided. Therefore, this criterion is not applied.
PP5 (Supporting)
According to standard ACMG guidelines, the rule for PP5 is: "Supporting Strength: Reputable source reports variant as pathogenic but evidence is not available for independent evaluation.". The evidence for this variant shows: ClinVar entries from three clinical labs report Pathogenic and one reports Likely Pathogenic. Therefore, this criterion is applied at Supporting strength.
BA1 (Not Applied)
According to VCEP guidelines, the rule for BA1 is: "Stand Alone Strength: gnomAD Filtering allele frequency >0.00056 (0.056%)". The evidence for this variant shows: absent from gnomAD. Therefore, this criterion is not applied.
BS1 (Not Applied)
According to VCEP guidelines, the rule for BS1 is: "Strong Strength: gnomAD Filtering allele frequency from 0.000043 up to 0.00056.". The evidence for this variant shows: absent from population databases. Therefore, this criterion is not applied.
BS2 (Not Applied)
According to VCEP guidelines, the rule for BS2 is: "Strong Strength: Observed homozygous in healthy individuals...". The evidence for this variant shows: no homozygous observations in unaffected individuals. Therefore, this criterion is not applied.
BS3 (Not Applied)
According to VCEP guidelines, the rule for BS3 is: "Strong Strength: Functional studies show no damaging effect.". The evidence for this variant shows: no such functional studies indicating no effect. Therefore, this criterion is not applied.
BS4 (Not Applied)
According to VCEP guidelines, the rule for BS4 is: "Strong Strength: Lack of segregation in two or more families.". The evidence for this variant shows: no segregation data. Therefore, this criterion is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, the rule for BP1 is: "Supporting Strength: Missense variant in gene where LOF is known mechanism.". The evidence for this variant shows: this is a truncating LOF variant. Therefore, this criterion is not applied.
BP2 (Not Applied)
According to VCEP guidelines, the rule for BP2 is: "Supporting Strength: Observed in trans with a pathogenic PTEN variant...". The evidence for this variant shows: no evidence of cis/trans observations with other variants. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule for BP3 is: "Supporting Strength: In-frame indels in a repetitive region.". The evidence for this variant shows: this is a nonsense variant. Therefore, this criterion is not applied.
BP4 (Not Applied)
According to VCEP guidelines, the rule for BP4 is: "Supporting Strength: Multiple lines of computational evidence suggest no impact on gene or gene product.". The evidence for this variant shows: despite benign computational scores, the truncating change itself is predicted to disrupt function. Therefore, this criterion is not applied.
BP5 (Not Applied)
According to VCEP guidelines, the rule for BP5 is: "Supporting Strength: Variant found in case with alternate molecular basis.". The evidence for this variant shows: no alternate molecular basis reported. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule for BP6 is: "Supporting Strength: Reputable source reports variant as benign but evidence not available.". The evidence for this variant shows: no benign entries in ClinVar. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to VCEP guidelines, the rule for BP7 is: "Supporting Strength: Synonymous or intronic variant with no splicing impact.". The evidence for this variant shows: this is a nonsense coding change. Therefore, this criterion is not applied.