Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_007294.2 | Alternative | 7191 nt | 201–5792 |
| NM_007294.3 | RefSeq Select | 7224 nt | 233–5824 |
| NM_007294.4 | MANE Select | 7088 nt | 114–5705 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenIARC class based on posterior probability from multifactorial likelihood analysis, thresholds for class as per Plon et al. 2008 (PMID: 18951446). Class 1 based on posterior probability = 0.000000000000233
This submission and the accompanying classification are no longer maintained by the submitter. For more information on current observations and classification, please contact variantquestions@myriad.com.
"This variant has been reported in ClinVar as Benign (9 clinical laboratories) and as Likely benign (4 clinical laboratories) and as Uncertain significance (1 clinical laboratories) and as Benign by Evidence-based Network for the Interpretation of Germline Mutant Alleles (ENIGMA) expert panel."
COSMIC Somatic Evidence
Open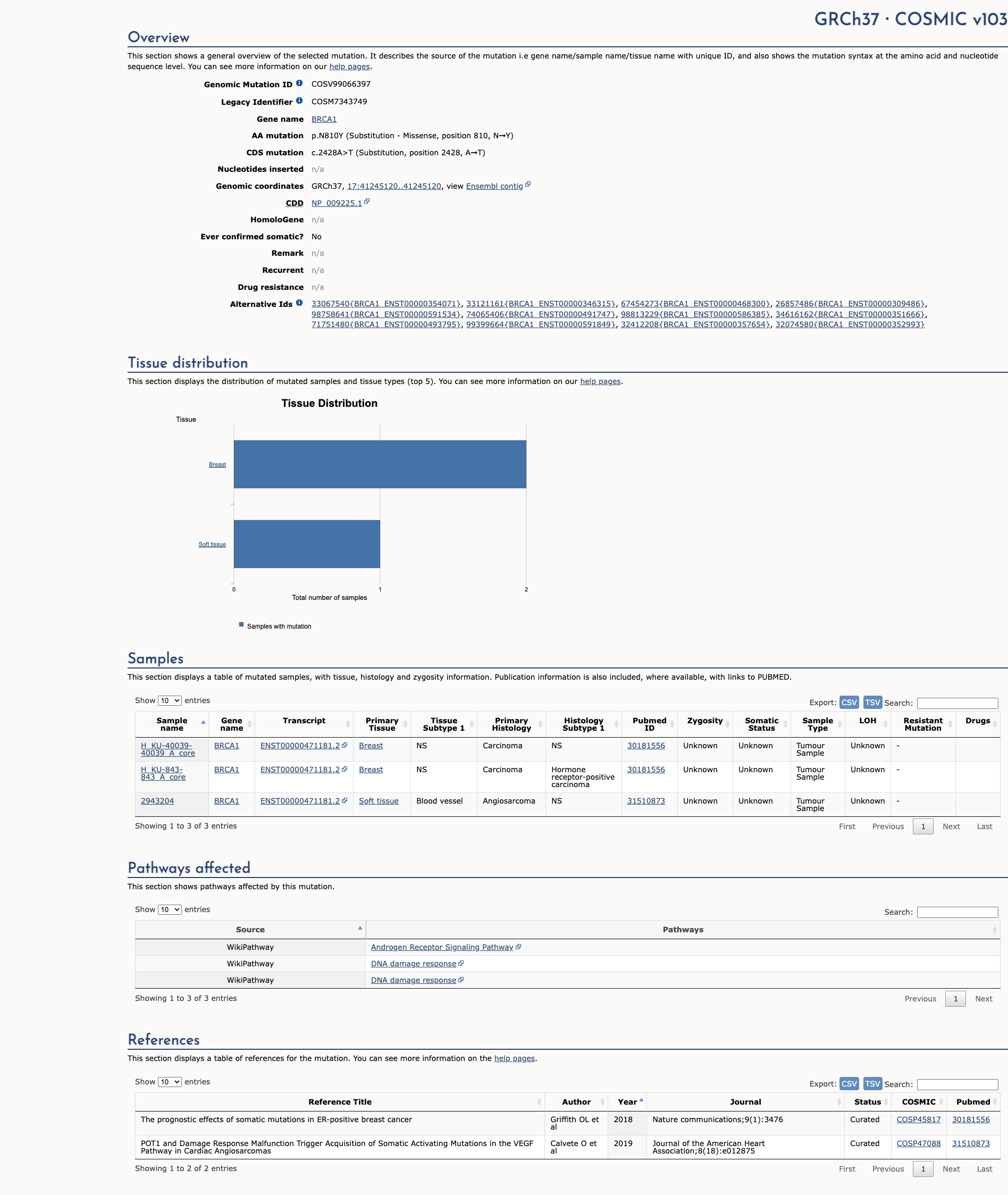
Functional Impact & Domains
Functional Domain
The BRCA1 N810Y variant has been functionally characterized and is likely neutral. In vitro studies using a cisplatin sensitivity assay indicate that this mutation does not significantly alter the function of the BRCA1 protein compared to the wildtype.
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | -199 bp |
| Donor Loss (DL) | 0.0 | -158 bp |
| Acceptor Gain (AG) | 0.0 | -355 bp |
| Donor Gain (DG) | 0.0 | 417 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Not Applied)
According to VCEP guidelines, "Very Strong Null variant (nonsense, frameshift, splice site (donor/acceptor +/-1,2), initiation codon, single or multi-exon deletion) in a gene where loss of function (LOF) is a known mechanism of disease." is the rule for PVS1. The evidence for this variant shows a missense substitution (p.N810Y), not a null variant. Therefore, this criterion is not applied.
PS1 (Not Applied)
According to VCEP guidelines, "Apply PS1 for predicted missense substitutions, where a previously classified pathogenic variant is considered to act via protein change (no confirmed or predicted effect on mRNA splicing (SpliceAI ≤0.1))." The evidence for this variant shows no previously established pathogenic variant at amino acid position 810. Therefore, this criterion is not applied.
PS2 (Not Applied)
According to standard ACMG guidelines, "De novo (both maternity and paternity confirmed) in a patient with the disease and no family history" is the rule for PS2. No evidence of de novo occurrence is provided for this variant. Therefore, this criterion is not applied.
PS3 (Not Applied)
According to VCEP guidelines, "Strong Well-established in vitro or in vivo functional studies supportive of a damaging effect" is the rule for PS3. The functional evidence shows no damaging effect in cisplatin sensitivity assays compared to wildtype. Therefore, this criterion is not applied.
PS4 (Not Applied)
According to VCEP guidelines, "Strong The prevalence of the variant in affected individuals is significantly increased compared to the prevalence in controls" is the rule for PS4. No case-control or affected individual data are available for p.N810Y. Therefore, this criterion is not applied.
PM1 (Not Applied)
According to VCEP guidelines, PM1 applies to variants located within a defined clinically important functional domain. The evidence shows p.N810Y is outside the RING (aa 2–101), coiled-coil (aa 1391–1424), and BRCT (aa 1650–1857) domains. Therefore, this criterion is not applied.
PM2 (Not Applied)
According to VCEP guidelines, "Supporting Absent from controls in an outbred population, from gnomAD v2.1 and v3.1" is the rule for PM2. The evidence shows the variant is present in gnomAD non-cancer exomes at MAF=0.00638%. Therefore, this criterion is not applied.
PM3 (Not Applied)
According to VCEP guidelines, PM3 applies to recessive co-occurrence in Fanconi anemia phenotypes. No such co-occurrence data are available for this variant. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, PM4 applies to protein length changes (in-frame indels or stop loss). The evidence shows p.N810Y is a missense substitution, not a length-altering variant. Therefore, this criterion is not applied.
PM5 (Not Applied)
According to VCEP guidelines, PM5 applies when a different proven pathogenic missense change at the same residue has been observed. There is no evidence of a known pathogenic variant at residue N810. Therefore, this criterion is not applied.
PM6 (Not Applied)
According to standard ACMG guidelines, PM6 applies to assumed de novo occurrences without confirmation. No such data are available for this variant. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to VCEP guidelines, PP1 applies to co-segregation with disease in multiple affected family members. No segregation data are provided for p.N810Y. Therefore, this criterion is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, "Missense variant in a gene with a low rate of benign missense variation and where missense is a common mechanism" is the rule for PP2. BRCA1 has a mix of benign and pathogenic missense variants, and no evidence supports a low benign missense rate here. Therefore, this criterion is not applied.
PP3 (Not Applied)
According to VCEP guidelines, PP3 applies to missense or in-frame variants inside a functional domain with predicted impact (BayesDel no-AF ≥0.28) or predicted splicing impact (SpliceAI ≥0.2). The evidence shows p.N810Y is outside all defined domains and SpliceAI=0. Therefore, this criterion is not applied.
PP4 (Not Applied)
According to VCEP guidelines, PP4 applies to multifactorial likelihood clinical data for breast cancer phenotype. No such data are available for p.N810Y. Therefore, this criterion is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, PP5 (reputable source declarations without evidence) is deprecated and should not be applied. Therefore, this criterion is not applied.
BA1 (Not Applied)
According to VCEP guidelines, "Stand Alone Filter allele frequency (FAF) is above 0.1%" is the rule for BA1. The evidence shows MAF=0.00638%, below this threshold. Therefore, this criterion is not applied.
BS1 (Supporting)
According to VCEP guidelines, "Filter allele frequency (FAF) is above 0.002% (FAF > 0.00002) and less than or equal to 0.01% (FAF ≤ 0.0001)" is the rule for BS1 Supporting. The evidence shows MAF=0.00638% in gnomAD non-cancer exomes, which falls within this range. Therefore, this criterion is applied at Supporting strength.
BS2 (Not Applied)
According to VCEP guidelines, BS2 applies to observations in healthy adults without a recessive phenotype. No such healthy adult data are available for this variant. Therefore, this criterion is not applied.
BS3 (Strong)
According to VCEP guidelines, "Strong Well-established in vitro or in vivo functional studies shows no damaging effect on protein function" is the rule for BS3. The evidence shows that cisplatin sensitivity assays indicate p.N810Y does not significantly alter BRCA1 function. Therefore, this criterion is applied at Strong strength.
BS4 (Not Applied)
According to VCEP guidelines, BS4 applies to lack of segregation in affected family members. No segregation analysis is provided. Therefore, this criterion is not applied.
BP1 (Strong)
According to VCEP guidelines, "Strong Apply BP1_Strong for silent substitution, missense or in-frame variants outside a clinically important functional domain AND no splicing predicted (SpliceAI ≤0.1)" is the rule for BP1. The evidence shows p.N810Y is outside the RING, coiled-coil, and BRCT domains and SpliceAI=0. Therefore, this criterion is applied at Strong strength.
BP2 (Not Applied)
According to standard ACMG guidelines, BP2 applies to observation in trans with a pathogenic variant for a dominant disorder or in cis with a pathogenic variant. No such co-observation data are available. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to VCEP guidelines, BP3 applies to in-frame indels in repetitive regions without functional impact. p.N810Y is a missense substitution, not an in-frame indel. Therefore, this criterion is not applied.
BP4 (Not Applied)
According to VCEP guidelines, BP4 applies to missense variants inside clinically important domains with benign computational predictions. p.N810Y is outside those domains and has mixed in silico results. Therefore, this criterion is not applied.
BP5 (Not Applied)
According to VCEP guidelines, BP5 applies to variants observed in a case with an alternate molecular basis for disease. No such data are available for this variant. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, BP6 (reputable source reports without evidence) is deprecated and should not be applied. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to VCEP guidelines, BP7 applies to silent or intronic variants without splicing impact. p.N810Y is a missense substitution. Therefore, this criterion is not applied.