Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_006218.2 | Alternative | 3724 nt | 158–3364 |
| NM_006218.3 | Alternative | 9104 nt | 158–3364 |
| NM_006218.4 | MANE Select | 9259 nt | 324–3530 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
Open""
COSMIC Somatic Evidence
Open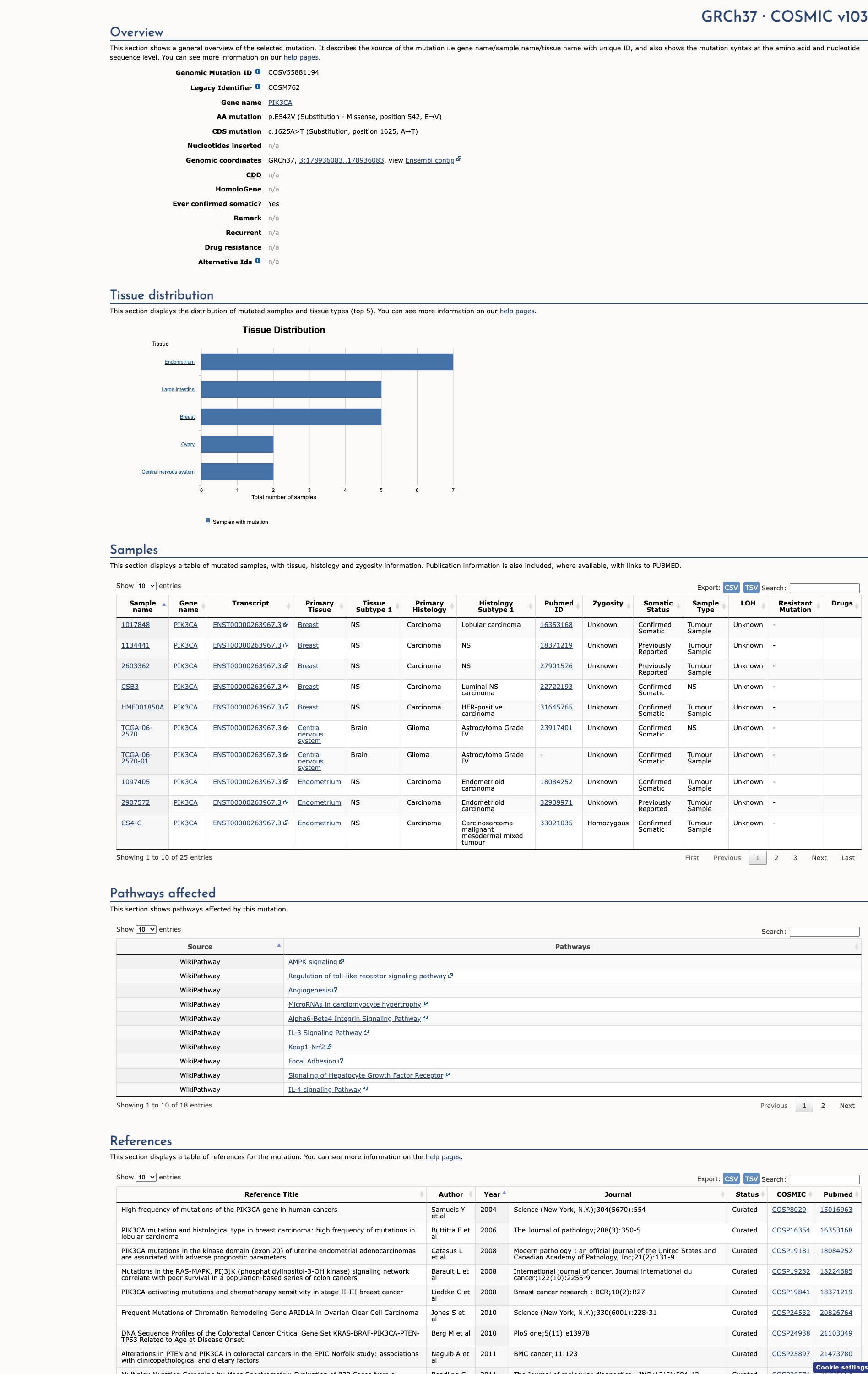
Functional Impact & Domains
Functional Domain
The PIK3CA E542V variant has been functionally characterized as oncogenic. It is located in the helical domain of the protein and confers a gain of function. Experimental evidence demonstrates that this mutation leads to increased downstream pathway activation, including enhanced phosphorylation of Akt and Mek1/2, and supports growth-factor independent cell survival. It is also transforming in cell culture models.
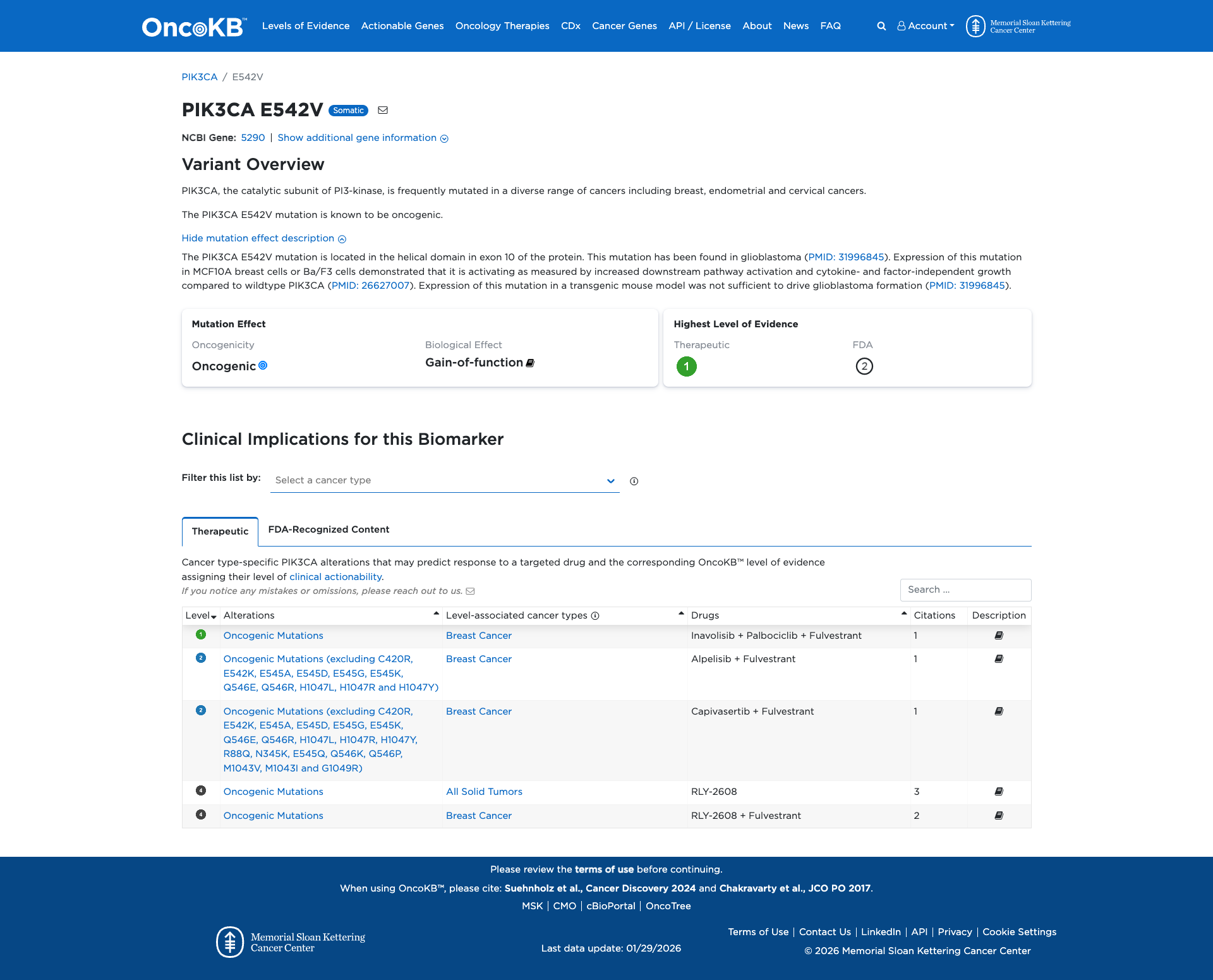
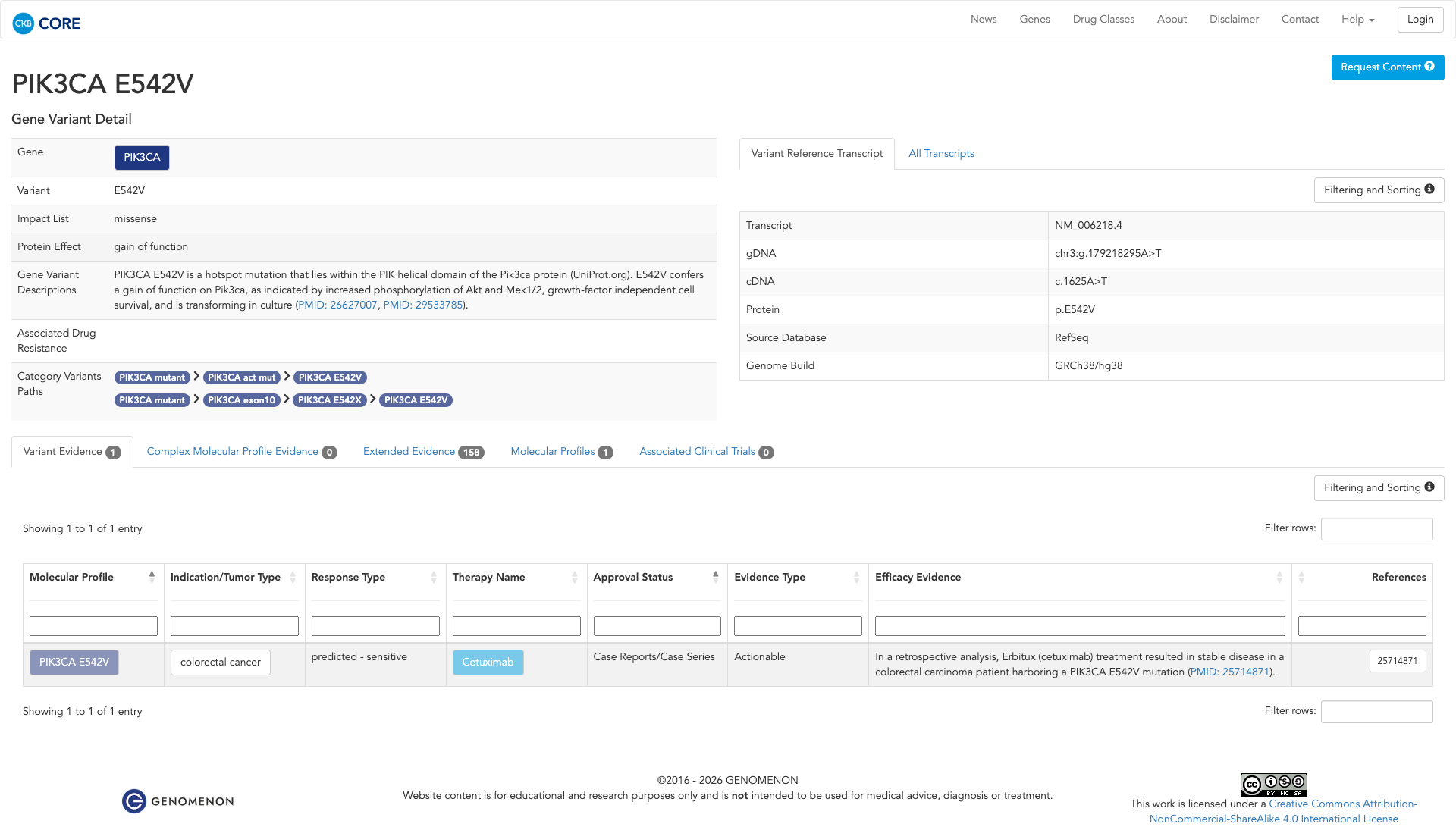
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | -85 bp |
| Donor Loss (DL) | 0.0 | 39 bp |
| Acceptor Gain (AG) | 0.01 | 14 bp |
| Donor Gain (DG) | 0.0 | 467 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Not Applied)
According to standard ACMG guidelines, the rule for PVS1 is: null variants (nonsense, frameshift, canonical ±1 or 2 splice sites, initiation codon, single‐ or multi‐exon deletion) in a gene where loss of function is a known mechanism of disease. The evidence for this variant shows: NM_006218.4:c.1625A>T is a missense change, not predicted to cause loss of function. Therefore, this criterion is not applied.
PS1 (Not Applied)
According to standard ACMG guidelines, the rule for PS1 is: same amino acid change as a previously established pathogenic variant regardless of nucleotide change. The evidence for this variant shows: E542V is a distinct amino acid change from the known pathogenic E542K. Therefore, this criterion is not applied.
PS2 (Not Applied)
According to VCEP guidelines, the rule for PS2 is: Strong if de novo with confirmed maternity/paternity and tissue evidence; Moderate if partially fulfilled. The evidence for this variant shows: no de novo or parental testing data available. Therefore, this criterion is not applied.
PS3 (Strong)
According to VCEP guidelines, the rule for PS3 is: Award PS3 if the functional assay meets the acceptability criteria delimited in (PMID: 31892348) with specifications added by the BMVCEP. The evidence for this variant shows: well‐established in vitro studies demonstrate gain‐of‐function in the helical domain with increased Akt/Mek1/2 phosphorylation and transforming activity. Therefore, this criterion is applied at Strong strength because the functional assays meet VCEP quality metrics supporting a damaging effect.
PS4 (Not Applied)
According to VCEP guidelines, the rule for PS4 is: points‐based case evidence only if variant meets PM2, with thresholds for Supporting, Moderate, Strong, Very Strong. The evidence for this variant shows: no published case series or phenotype scoring available. Therefore, this criterion is not applied.
PM1 (Supporting)
According to VCEP guidelines, the rule for PM1 is: Residues affecting critical functional domains provided in Table 4 for each gene (Supporting strength). The evidence for this variant shows: E542 is in the well‐established helical domain mutational hotspot of PIK3CA. Therefore, this criterion is applied at Supporting strength because it lies in a critical functional domain without benign variation.
PM2 (Supporting)
According to VCEP guidelines, the rule for PM2 is: Absent/rare from controls in an ethnically‐matched cohort population sample (Supporting strength). The evidence for this variant shows: not found in gnomAD (MAF 0%). Therefore, this criterion is applied at Supporting strength because it is absent from population controls.
PM3 (Not Applied)
According to standard ACMG guidelines, the rule for PM3 is: for recessive disorders, detection in trans with a pathogenic variant. The evidence for this variant shows: PIK3CA E542V is not studied in a recessive context and no trans data. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, the rule for PM4 is: protein length changes due to in‐frame indels or stop‐loss. The evidence for this variant shows: E542V is a missense change without protein length alteration. Therefore, this criterion is not applied.
PM5 (Moderate)
According to standard ACMG guidelines, the rule for PM5 is: novel missense change at an amino acid residue where a different pathogenic missense change has been observed (Moderate strength). The evidence for this variant shows: E542K is a well‐established pathogenic variant at the same residue. Therefore, this criterion is applied at Moderate strength because this is a novel change at a codon with known pathogenic variation.
PM6 (Not Applied)
According to standard ACMG guidelines, the rule for PM6 is: assumed de novo without confirmation. The evidence for this variant shows: no de novo data. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to standard ACMG guidelines, the rule for PP1 is: cosegregation with disease in multiple affected family members. The evidence for this variant shows: no family segregation data. Therefore, this criterion is not applied.
PP2 (Supporting)
According to VCEP guidelines, the rule for PP2 is: missense constraint computed in ExAC/gnomAD with z‐score >3.09 applicable to PIK3CA (Supporting strength). The evidence for this variant shows: PIK3CA exhibits high missense constraint (z‐score >3.09). Therefore, this criterion is applied at Supporting strength because the gene is constrained for missense variation.
PP3 (Not Applied)
According to standard ACMG guidelines, the rule for PP3 is: multiple lines of computational evidence support a deleterious effect. The evidence for this variant shows: mixed in silico predictions (some tools benign, some pathogenic). Therefore, this criterion is not applied.
PP4 (Not Applied)
According to standard ACMG guidelines, the rule for PP4 is: patient’s phenotype or family history is highly specific for a disease with a single genetic etiology. The evidence for this variant shows: no clinical phenotype or family history provided. Therefore, this criterion is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, the rule for PP5 is: reputable source reports the variant as pathogenic without available evidence. The evidence for this variant shows: not present in ClinVar or other databases. Therefore, this criterion is not applied.
BA1 (Not Applied)
According to VCEP guidelines, the rule for BA1 is: allele frequency >0.0926% in general population (Stand‐alone). The evidence for this variant shows: MAF 0% in gnomAD. Therefore, this criterion is not applied.
BS1 (Not Applied)
According to VCEP guidelines, the rule for BS1 is: allele frequency >0.0185% in population (Strong). The evidence for this variant shows: MAF 0% in gnomAD. Therefore, this criterion is not applied.
BS2 (Not Applied)
According to VCEP guidelines, the rule for BS2 is: ≥3 homozygotes or well‐phenotyped heterozygotes in population (Strong). The evidence for this variant shows: no homozygotes or reported heterozygotes. Therefore, this criterion is not applied.
BS3 (Not Applied)
According to VCEP guidelines, the rule for BS3 is: well‐established functional studies show no damaging effect (Strong/Supporting). The evidence for this variant shows: functional studies show a damaging gain‐of‐function effect. Therefore, this criterion is not applied.
BS4 (Not Applied)
According to standard ACMG guidelines, the rule for BS4 is: lack of segregation in affected members of a family. The evidence for this variant shows: no segregation data. Therefore, this criterion is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, the rule for BP1 is: missense variant in gene where only truncating variants cause disease. The evidence for this variant shows: PIK3CA is an oncogene driven by missense gain‐of‐function. Therefore, this criterion is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, the rule for BP2 is: observed in trans with a pathogenic variant for a recessive disorder. The evidence for this variant shows: no trans data or recessive context. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule for BP3 is: in‐frame indels in repetitive regions without functional impact. The evidence for this variant shows: missense, not an indel. Therefore, this criterion is not applied.
BP4 (Not Applied)
According to VCEP guidelines, the rule for BP4 is: applicable only to synonymous, intronic (except canonical splice sites), or UTR variants if splicing tools predict no impact. The evidence for this variant shows: missense change. Therefore, this criterion is not applied.
BP5 (Not Applied)
According to standard ACMG guidelines, the rule for BP5 is: variant found in a case with an alternate molecular basis for disease. The evidence for this variant shows: no such data. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule for BP6 is: reputable source reports variant as benign without evidence. The evidence for this variant shows: no database entries. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to VCEP guidelines, the rule for BP7 is: synonymous or intronic variants with low conservation. The evidence for this variant shows: missense variant. Therefore, this criterion is not applied.