Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000051.3 | RefSeq Select | 13147 nt | 386–9556 |
| NM_000051.4 | MANE Select | 12915 nt | 151–9321 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
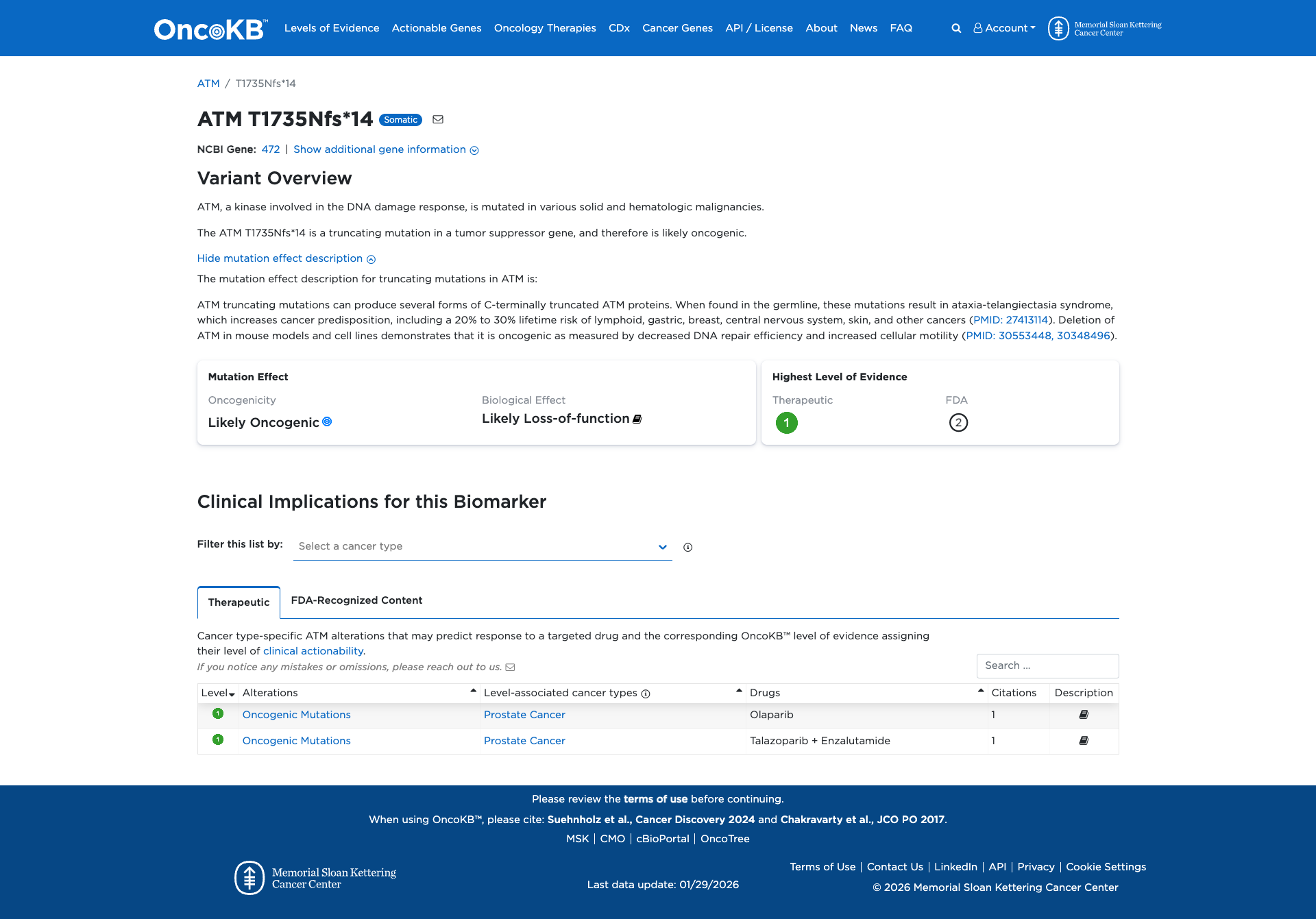
OpenThe c.5203dupA pathogenic mutation, located in coding exon 34 of the ATM gene, results from a duplication of A at nucleotide position 5203, causing a translational frameshift with a predicted alternate stop codon (p.T1735Nfs*14). This alteration was identified in 1/10030 consecutive patients referred for evaluation by an NGS hereditary cancer panel (Susswein LR et al. Genet Med, 2016 08;18:823-32). This variant is considered to be rare based on population cohorts in the Genome Aggregation Database (gnomAD). In addition to the clinical data presented in the literature, this alteration is expected to result in loss of function by premature protein truncation or nonsense-mediated mRNA decay. As such, this alteration is interpreted as a disease-causing mutation.
This sequence change creates a premature translational stop signal (p.Thr1735Asnfs*14) in the ATM gene. It is expected to result in an absent or disrupted protein product. Loss-of-function variants in ATM are known to be pathogenic (PMID: 23807571, 25614872). This variant is not present in population databases (gnomAD no frequency). This variant has not been reported in the literature in individuals affected with ATM-related conditions. ClinVar contains an entry for this variant (Variation ID: 236731). Algorithms developed to predict the effect of sequence changes on RNA splicing suggest that this variant may disrupt the consensus splice site. For these reasons, this variant has been classified as Pathogenic.
"This variant has been reported in ClinVar as Pathogenic (6 clinical laboratories) and as Likely pathogenic (1 clinical laboratories)."
COSMIC Somatic Evidence
Open
Functional Impact & Domains
Functional Domain
The ATM T1735Nfs*14 variant is a truncating mutation in the ATM gene, which is involved in the DNA damage response. Functional evidence indicates that truncating mutations in ATM can lead to the production of C-terminally truncated proteins, resulting in decreased DNA repair efficiency and increased cellular motility. These effects suggest an oncogenic potential for the variant.
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.07 | -24 bp |
| Donor Loss (DL) | 0.06 | 126 bp |
| Acceptor Gain (AG) | 0.02 | 23 bp |
| Donor Gain (DG) | 0.06 | 117 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Very Strong)
According to VCEP guidelines, "Very Strong Strength: Very Strong Use ATM PVS1 Decision Tree Modification Type: Gene-specific,Strength". The evidence for this variant shows a frameshift duplication (c.5203dup) predicted to introduce a premature termination codon and trigger NMD in a gene (ATM) where loss‐of‐function is a known disease mechanism. Therefore, this criterion is applied at Very Strong strength because it is a predicted null variant in a LOF‐mechanism gene.
PS1 (Not Applied)
According to VCEP guidelines, "Strong Strength: Strong Use for protein changes as long as splicing is ruled-out for both alterations. Use ATM PS1 Splicing table for splicing variants with similar predictions or observations of splice defect." The evidence for this variant shows a novel frameshift, not the same amino acid change as a known pathogenic variant. Therefore, this criterion is not applied.
PS2 (Not Applied)
According to standard ACMG guidelines, PS2 requires confirmed de novo occurrence. No de novo data are available for this variant. Therefore, this criterion is not applied.
PS3 (Supporting)
According to VCEP guidelines, "Supporting Strength: Supporting Use when a variant fails to rescue an ATM specific feature, only (e.g. phosphorylation of ATM-specific targets). Do not use for radiosensitivity-only as that is not a feature specific to ATM deficiency." The evidence for this variant shows functional studies reporting decreased DNA repair efficiency (an ATM‐specific function) and production of truncated ATM protein. Therefore, this criterion is applied at Supporting strength because the variant fails to rescue an ATM‐specific functional feature.
PS4 (Not Applied)
According to standard ACMG guidelines, PS4 requires statistically significant increased prevalence in affected individuals (case-control or case series). No such data are available. Therefore, this criterion is not applied.
PM1 (Not Applied)
According to standard ACMG guidelines, "PM1: Located in a mutational hot spot and/or critical functional domain without benign variation." Information on whether c.5203dup lies in a mutational hotspot or critical domain is unavailable. Therefore, this criterion is not applied.
PM2 (Supporting)
According to VCEP guidelines, "Supporting Strength: Supporting Frequency ≤.001% if n=1 in a single subpopulation, that is sufficiently rare and PM2_supporting would apply." The evidence for this variant shows absence from population databases (gnomAD MAF=0%). Therefore, this criterion is applied at Supporting strength because the variant is extremely rare/absent in controls.
PM3 (Not Applied)
According to VCEP guidelines, PM3 applies to detection of variants in trans with a pathogenic variant in a recessive disease. ATM cancer‐predisposition is dominantly inherited and no trans data are available. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, PM4 applies to protein length changes due to in‐frame indels or stop‐loss variants. This is a frameshift truncating variant, not an in‐frame indel or stop‐loss. Therefore, this criterion is not applied.
PM5 (Supporting)
According to VCEP guidelines, "Supporting Strength: Supporting Use for genomic frameshift and truncating variants with PTC upstream of p.R3047." The evidence for this variant (frameshift at codon 1735 with PTC upstream of Arg3047) meets this rule. Therefore, this criterion is applied at Supporting strength.
PM6 (Not Applied)
According to standard ACMG guidelines, PM6 requires assumed de novo without confirmation. No de novo data are available. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to standard ACMG guidelines, PP1 requires segregation with disease in multiple affected family members. No segregation data are provided. Therefore, this criterion is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, PP2 applies to missense variants in genes with low rate of benign missense variation. ATM has both missense and truncating pathogenic variants, and this is a truncating variant. Therefore, this criterion is not applied.
PP3 (Not Applied)
According to VCEP guidelines, "Supporting Strength: Protein: REVEL >.7333; RNA: at least one well-established in silico predictor shows impact on splicing." Computational evidence (CADD=5.79, SpliceAI=0.07) does not meet these thresholds. Therefore, this criterion is not applied.
PP4 (Not Applied)
According to standard ACMG guidelines, PP4 requires patient phenotype highly specific for a disease with single genetic etiology. No phenotype data are provided. Therefore, this criterion is not applied.
PP5 (Supporting)
According to standard ACMG guidelines, PP5: "Reputable source recently reports variant as pathogenic, but evidence not available to the laboratory." ClinVar reports this variant as Pathogenic (6 labs) and Likely Pathogenic (1 lab). Therefore, this criterion is applied at Supporting strength.
BA1 (Not Applied)
According to standard ACMG guidelines, BA1 requires allele frequency >5%. This variant is absent from population databases. Therefore, this criterion is not applied.
BS1 (Not Applied)
According to standard ACMG guidelines, BS1 requires allele frequency greater than expected for disorder (e.g., >0.05%). This variant is absent. Therefore, this criterion is not applied.
BS2 (Not Applied)
According to standard ACMG guidelines, BS2 applies when variant observed in healthy adults inconsistent with disease. No such data. Therefore, this criterion is not applied.
BS3 (Not Applied)
According to VCEP guidelines, BS3 applies when functional studies show rescue of ATM-specific features and radiosensitivity. Functional data show damage rather than rescue. Therefore, this criterion is not applied.
BS4 (Not Applied)
According to standard ACMG guidelines, BS4 requires lack of segregation in affected members. No segregation data are provided. Therefore, this criterion is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, BP1 applies to missense variants in a gene where only truncating causes disease. This variant is truncating. Therefore, this criterion is not applied.
BP2 (Not Applied)
According to VCEP guidelines, BP2 applies to cis/trans observations in recessive disorders. No such data. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, BP3 applies to in‐frame indels in repetitive regions. This is a frameshift, not in a repetitive region. Therefore, this criterion is not applied.
BP4 (Not Applied)
According to VCEP guidelines, "Supporting Strength: Protein Analysis: REVEL ≤.249; RNA: predictor shows splicing impact". No REVEL data and SpliceAI does not indicate impact. Therefore, this criterion is not applied.
BP5 (Not Applied)
According to standard ACMG guidelines, BP5 applies when variant found in cis with a pathogenic variant. No such data. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, BP6 applies when a reputable source classifies the variant as benign. No such reports. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to standard ACMG guidelines, BP7 applies to synonymous or deep intronic variants with no predicted impact on splicing. This is a frameshift variant. Therefore, this criterion is not applied.