Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_001754.3 | Alternative | 6190 nt | 400–1842 |
| NM_001754.4 | RefSeq Select | 5967 nt | 191–1633 |
| NM_001754.5 | MANE Select | 5971 nt | 195–1637 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
Open""
COSMIC Somatic Evidence
Open
Functional Impact & Domains
Functional Domain
The RUNX1 F158Lfs*13 variant is a truncating mutation in the RUNX1 gene, a tumor suppressor involved in hematopoietic differentiation. Functional evidence indicates that truncating mutations in RUNX1 typically inhibit its function, contributing to oncogenesis and predisposing individuals to hematologic malignancies such as leukemia, lymphomas, and myelodysplastic syndromes.
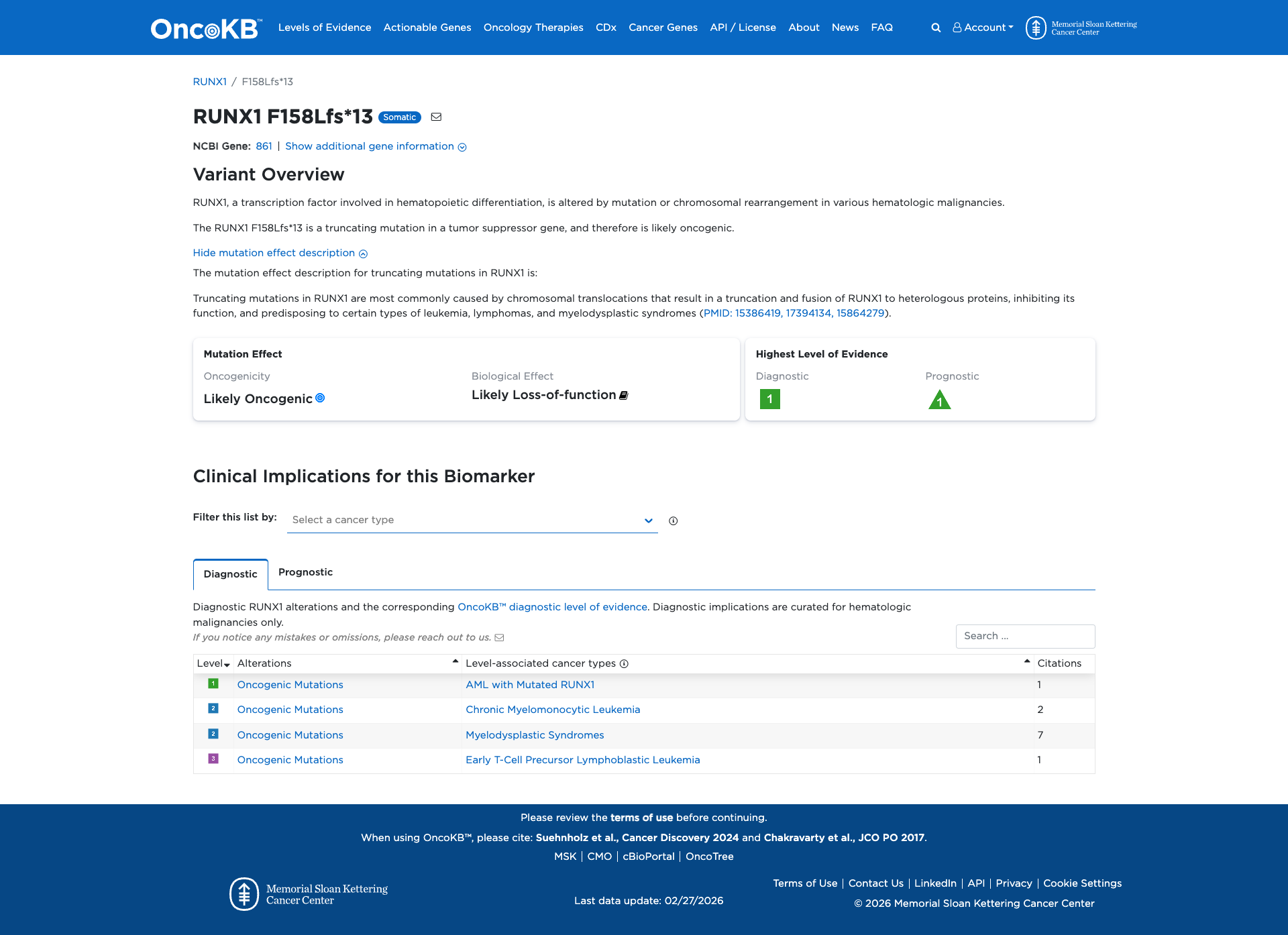
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | 4 bp |
| Donor Loss (DL) | 0.18 | 5 bp |
| Acceptor Gain (AG) | 0.0 | 138 bp |
| Donor Gain (DG) | 0.04 | 465 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Very Strong)
According to VCEP guidelines, the rule for PVS1 is: "Very Strong Strength: Very Strong Per modified RUNX1 PVS1 decision tree for SNVs and CNVs and table of splicing effects." The evidence for this variant shows: NM_001754.4:c.474_489delTAATGACCTCAGGTTT (F158Lfs*13) is a frameshift truncating variant upstream of the last exon in RUNX1, a gene where loss of function is a known mechanism of disease. Therefore, this criterion is applied at Very Strong strength because it meets the VCEP PVS1 rule for a null variant in RUNX1.
PS1 (Not Applied)
According to standard ACMG guidelines, the rule for PS1 is: "Same amino acid change as a previously established pathogenic variant regardless of nucleotide change." The evidence for this variant shows: F158Lfs*13 is a novel frameshift, not a recurrent amino acid substitution. Therefore, PS1 is not applied.
PS2 (Not Applied)
According to standard ACMG guidelines, the rule for PS2 is: "De novo (both maternity and paternity confirmed) in a patient with the disease and no family history." The evidence for this variant shows: no de novo status or parental testing data available. Therefore, PS2 is not applied.
PS3 (Not Applied)
According to VCEP guidelines, the rule for PS3 is: "Strong Transactivation assays demonstrating altered transactivation (<20% of wt) AND data from a secondary assay demonstrating altered function. Not applicable if variant meets PVS1. If variant meets PVS1_strong, either apply PS3_moderate or upgrade to PVS1." The evidence for this variant shows: it meets PVS1. Therefore, PS3 is not applied.
PS4 (Not Applied)
According to VCEP guidelines, the rule for PS4 is: "Strong: ≥4 probands meeting at least one of the RUNX1-phenotypic criteria; Moderate: 2-3 probands; Supporting: 1 proband." The evidence for this variant shows: no case-level proband data reported. Therefore, PS4 is not applied.
PM1 (Not Applied)
According to VCEP guidelines, the rule for PM1 is: "Supporting Strength: Variant affecting one of the other amino acid residues 89-204 within the RHD (excluding key residues)." The evidence for this variant shows: it is a frameshift deletion, not a missense/in-frame alteration at a specific RHD residue. Therefore, PM1 is not applied.
PM2 (Supporting)
According to VCEP guidelines, the rule for PM2 is: "Supporting Strength: Variant must be completely absent from all population databases." The evidence for this variant shows: it is absent from gnomAD and other population datasets. Therefore, PM2 is applied at Supporting strength because the variant is absent from population databases.
PM3 (Not Applied)
According to standard ACMG guidelines, the rule for PM3 is: "For recessive disorders, detected in trans with a pathogenic variant." The evidence for this variant shows: no evidence of trans phase or recessive inheritance. Therefore, PM3 is not applied.
PM4 (Not Applied)
According to VCEP guidelines, the rule for PM4 is: "Moderate Strength: In-frame deletion/insertion impacting key RHD residues; Supporting Strength: in-frame deletion/insertion impacting other RHD residues." The evidence for this variant shows: it is a frameshift, not an in-frame indel. Therefore, PM4 is not applied.
PM5 (Supporting)
According to VCEP guidelines, the rule for PM5 is: "Supporting Strength: PM5_supporting is also applied to nonsense/frameshift variants that are downstream of c.98." The evidence for this variant shows: F158Lfs*13 is a frameshift truncation downstream of c.98. Therefore, PM5 is applied at Supporting strength.
PM6 (Not Applied)
According to standard ACMG guidelines, the rule for PM6 is: "Assumed de novo, but without confirmation of paternity and maternity." The evidence for this variant shows: no de novo data available. Therefore, PM6 is not applied.
PP1 (Not Applied)
According to VCEP guidelines, the rule for PP1 is: "Supporting Strength: 3-4 meioses co-segregation; Moderate: 5-6 meioses; Strong: ≥7 meioses." The evidence for this variant shows: no family segregation data. Therefore, PP1 is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, the rule for PP2 is: "Missense variant in a gene with low rate of benign missense variation and where missense is a common mechanism of disease." The evidence for this variant shows: it is a frameshift, not a missense. Therefore, PP2 is not applied.
PP3 (Not Applied)
According to VCEP guidelines, the rule for PP3 is: "Supporting Strength: For missense variants, REVEL ≥0.88; for splice, SpliceAI ≥0.38." The evidence for this variant shows: it is a frameshift, and in silico splicing impact is below threshold. Therefore, PP3 is not applied.
PP4 (Not Applied)
According to standard ACMG guidelines, the rule for PP4 is: "Patient’s phenotype or family history is highly specific for a disease with a single genetic etiology." The evidence for this variant shows: no detailed phenotypic specificity provided. Therefore, PP4 is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, the rule for PP5 is: "Reputable source reports variant as pathogenic without evidence available." The evidence for this variant shows: not reported in ClinVar or other databases. Therefore, PP5 is not applied.
BA1 (Not Applied)
According to VCEP guidelines, the rule for BA1 is: "Stand Alone Strength: Minor allele frequency ≥0.15%." The evidence for this variant shows: MAF = 0%. Therefore, BA1 is not applied.
BS1 (Not Applied)
According to VCEP guidelines, the rule for BS1 is: "Strong Strength: MAF between 0.015% and 0.15%." The evidence for this variant shows: absent from population databases. Therefore, BS1 is not applied.
BS2 (Not Applied)
According to standard ACMG guidelines, the rule for BS2 is: "Observed in a healthy adult individual for a fully penetrant dominant disorder." The evidence for this variant shows: no healthy individuals reported. Therefore, BS2 is not applied.
BS3 (Not Applied)
According to VCEP guidelines, the rule for BS3 is: "Strong Strength: Transactivation assays demonstrating normal function; Supporting Strength: normal transactivation only." The evidence for this variant shows: functional data indicate loss of function. Therefore, BS3 is not applied.
BS4 (Not Applied)
According to standard ACMG guidelines, the rule for BS4 is: "Lack of segregation in affected members of a family." The evidence for this variant shows: no segregation data available. Therefore, BS4 is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, the rule for BP1 is: "Missense variant in a gene where only truncating variants cause disease." The evidence for this variant shows: it is truncating. Therefore, BP1 is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, the rule for BP2 is: "Observed in trans with a pathogenic variant for a dominant gene or in cis with a pathogenic variant." The evidence for this variant shows: no such data. Therefore, BP2 is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule for BP3 is: "In-frame indel in a repetitive region without a known function." The evidence for this variant shows: it is a frameshift not in a repetitive region. Therefore, BP3 is not applied.
BP4 (Not Applied)
According to VCEP guidelines, the rule for BP4 is: "Supporting Strength: REVEL <0.50 and SpliceAI ≤0.20 for missense/synonymous/intronic." The evidence for this variant shows: it is a frameshift. Therefore, BP4 is not applied.
BP5 (Not Applied)
According to standard ACMG guidelines, the rule for BP5 is: "Variant found in a case with an alternate molecular basis for disease." The evidence for this variant shows: no alternate molecular basis reported. Therefore, BP5 is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule for BP6 is: "Reputable source reports variant as benign without evidence." The evidence for this variant shows: no such report. Therefore, BP6 is not applied.
BP7 (Not Applied)
According to VCEP guidelines, the rule for BP7 is: "Supporting Strength: Synonymous or intronic variant with SpliceAI ≤0.20 and phyloP ≤2.0." The evidence for this variant shows: it is a frameshift. Therefore, BP7 is not applied.