Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000051.3 | RefSeq Select | 13147 nt | 386–9556 |
| NM_000051.4 | MANE Select | 12915 nt | 151–9321 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThis variant is considered likely benign. This variant is strongly associated with less severe personal and family histories of cancer, typical for individuals without pathogenic variants in this gene [PMID: 25085752].
"This variant has been reported in ClinVar as Uncertain significance (8 clinical laboratories) and as Likely benign (1 clinical laboratories)."
COSMIC Somatic Evidence
Open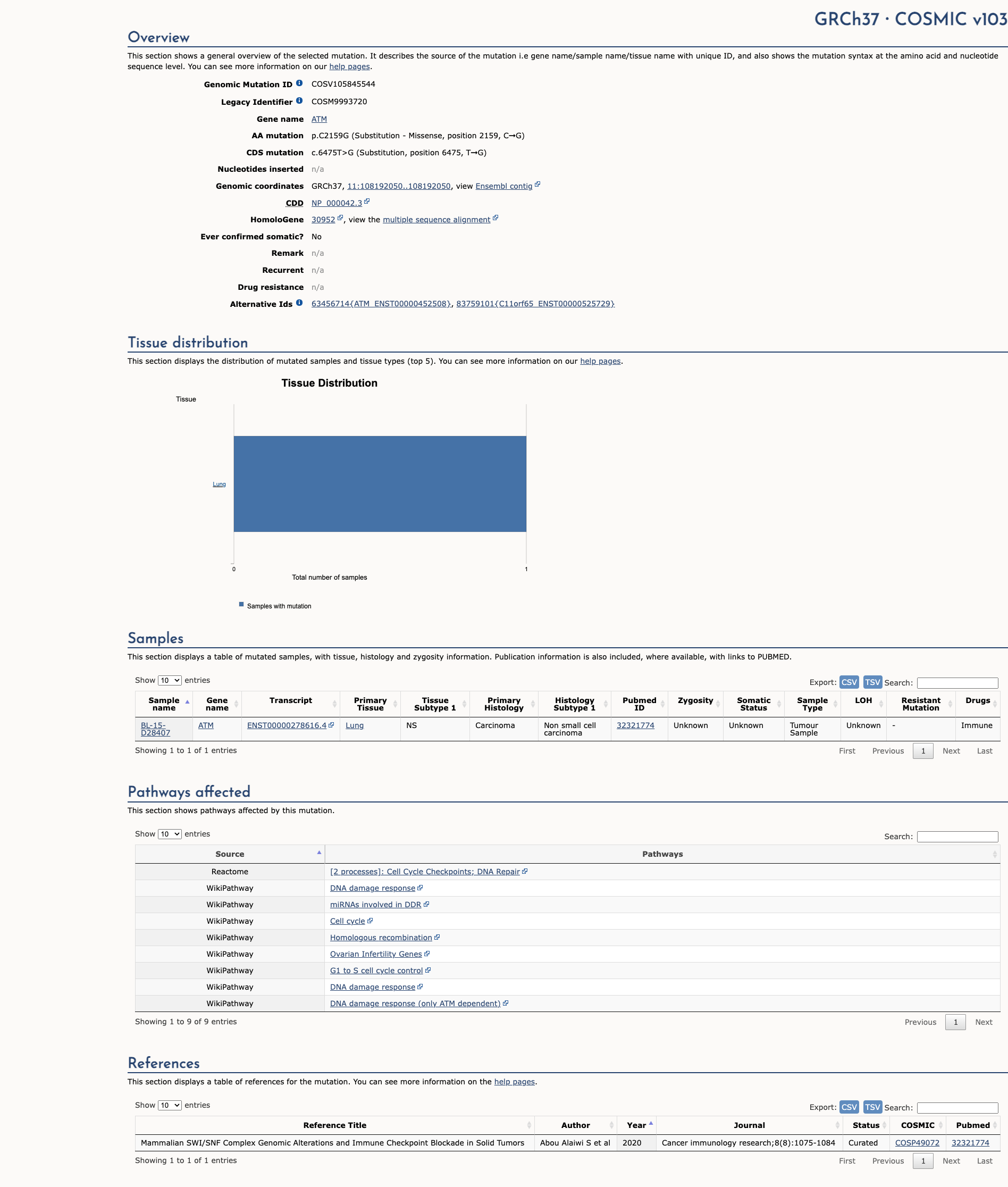
Functional Impact & Domains
Functional Domain
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.04 | -20 bp |
| Donor Loss (DL) | 0.02 | 97 bp |
| Acceptor Gain (AG) | 0.12 | 47 bp |
| Donor Gain (DG) | 0.13 | 0 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Not Applied)
According to VCEP guidelines: “Use ATM PVS1 Decision Tree” which applies to null variants causing loss of function. The evidence for this variant shows it is a missense change (Cys2159Gly), not a predicted null allele. Therefore, this criterion is not applied because PVS1 is reserved for LOF variants and this is a missense.
PS1 (Not Applied)
According to VCEP guidelines: “Use for protein changes as long as splicing is ruled‐out for both alterations.” The evidence shows no previously established pathogenic variant resulting in Cys2159Gly. Therefore, PS1 is not applied because there is no existing pathogenic variant with the same amino acid change.
PS2 (Not Applied)
According to standard ACMG guidelines: PS2 requires confirmed de novo occurrence. The evidence for this variant shows no information on parental testing or de novo status. Therefore, PS2 is not applied due to lack of de novo confirmation data.
PS3 (Not Applied)
According to VCEP guidelines: PS3 requires functional studies showing failure to rescue ATM‐specific features. The evidence shows no functional characterization for Cys2159Gly. Therefore, PS3 is not applied because no functional data are available.
PS4 (Not Applied)
According to standard ACMG guidelines: PS4 requires statistically significant case‐control data or proband counting. There are no published case‐control studies or sufficient proband data for this variant. Therefore, PS4 is not applied.
PM1 (Not Applied)
According to standard ACMG guidelines: PM1 applies to variants in mutational hot spots or critical functional domains. There is no evidence that Cys2159 lies within a defined hotspot or critical domain. Therefore, PM1 is not applied.
PM2 (Not Applied)
According to VCEP guidelines: “Frequency ≤0.001% if n=1 in a single subpopulation” qualifies for PM2_supporting. The evidence shows MAF=0.00283% with n=8 alleles. Therefore, PM2 is not applied because the allele count and frequency exceed the VCEP‐specified threshold.
PM3 (Not Applied)
According to VCEP guidelines: PM3 applies to recessive disorders when in trans with a pathogenic variant. There is no evidence of trans phase with a known pathogenic ATM variant. Therefore, PM3 is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines: PM4 applies to protein length changes (in‐frame indels or stop‐loss). This is a missense change without length alteration. Therefore, PM4 is not applied.
PM5 (Not Applied)
According to VCEP guidelines: PM5_supporting applies when a novel missense occurs at a residue with a different known pathogenic missense. No other pathogenic variant at residue 2159 is reported. Therefore, PM5 is not applied.
PM6 (Not Applied)
According to standard ACMG guidelines: PM6 applies to presumed de novo variants without parental confirmation. No de novo assumption has been made. Therefore, PM6 is not applied.
PP1 (Not Applied)
According to standard ACMG guidelines: PP1 requires cosegregation with disease in multiple affected family members. No segregation data are available. Therefore, PP1 is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines: PP2 applies to missense variants in genes with low benign missense variation. ATM tolerates missense variation and has many benign/transitional variants. Therefore, PP2 is not applied.
PP3 (Not Applied)
According to VCEP guidelines: “PP3 supporting if REVEL >0.7333 or splicing predictor shows impact.” The evidence shows REVEL=0.39 and SpliceAI Δscore=0.13, indicating no predicted impact. Therefore, PP3 is not applied.
PP4 (Not Applied)
According to standard ACMG guidelines: PP4 requires a highly specific phenotype or family history. No phenotype data are provided. Therefore, PP4 is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines: PP5 applies when a reputable source classifies the variant as pathogenic. ClinVar reports VUS or Likely benign. Therefore, PP5 is not applied.
BA1 (Not Applied)
According to VCEP guidelines: BA1 stand‐alone if allele frequency >0.5%. The observed frequency is 0.00283%. Therefore, BA1 is not applied.
BS1 (Not Applied)
According to VCEP guidelines: BS1 strong if allele frequency >5%. The observed frequency is 0.00283%. Therefore, BS1 is not applied.
BS2 (Not Applied)
According to standard ACMG guidelines: BS2 applies when variant observed in healthy adults inconsistent with disease penetrance. No such observations are reported. Therefore, BS2 is not applied.
BS3 (Not Applied)
According to VCEP guidelines: BS3 requires functional evidence of rescue of ATM activity. No functional rescue data exist. Therefore, BS3 is not applied.
BS4 (Not Applied)
According to standard ACMG guidelines: BS4 requires lack of segregation in affected family members. No such data are provided. Therefore, BS4 is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines: BP1 applies to missense in genes where only truncating variants cause disease. ATM has known pathogenic missense variants. Therefore, BP1 is not applied.
BP2 (Not Applied)
According to VCEP guidelines: BP2 refers to observed in cis/trans with pathogenic variant inconsistent with disease mechanism. No such data exist. Therefore, BP2 is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines: BP3 applies to in‐frame indels in repetitive regions. This is not an indel. Therefore, BP3 is not applied.
BP4 (Not Applied)
According to VCEP guidelines: BP4 supporting requires REVEL ≤0.249 or benign splicing prediction. REVEL=0.39 exceeds threshold; splicing Δscore=0.13 is low but RNA thresholds are not defined for benign. Therefore, BP4 is not applied.
BP5 (Not Applied)
According to standard ACMG guidelines: BP5 applies when variant found in trans with a pathogenic variant for a fully penetrant disorder. No such trans configuration is known. Therefore, BP5 is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines: BP6 applies when a reputable source classifies as benign without primary data. ClinVar reports conflicting VUS/Likely benign but not unanimously benign. Therefore, BP6 is not applied.
BP7 (Not Applied)
According to standard ACMG guidelines: BP7 applies to synonymous or deep intronic variants. This is a missense change. Therefore, BP7 is not applied.