Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000251.2 | RefSeq Select | 3226 nt | 126–2930 |
| NM_000251.1 | Alternative | 3145 nt | 69–2873 |
| NM_000251.3 | MANE Select | 3115 nt | 37–2841 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThe c.1601delG pathogenic mutation, located in coding exon 10 of the MSH2 gene, results from a deletion of one nucleotide at nucleotide position 1601, causing a translational frameshift with a predicted alternate stop codon (p.R534Lfs*9). This mutation was observed as a somatic alteration in the colon tumor of a 55-year-old woman who tested positive for a different germline MSH2 mutation; the colon tumor demonstrated microsatellite instability and loss of MSH2 and MSH6 on immunohistochemical staining (Vargas-Parra GM et al. Int. J. Cancer 2017 10;141(7):1365-1380). In addition to the clinical data presented in the literature, this alteration is expected to result in loss of function by premature protein truncation or nonsense-mediated mRNA decay. As such, this alteration is interpreted as a disease-causing mutation.
"This variant has been reported in ClinVar as Pathogenic (1 clinical laboratories)."
COSMIC Somatic Evidence
Open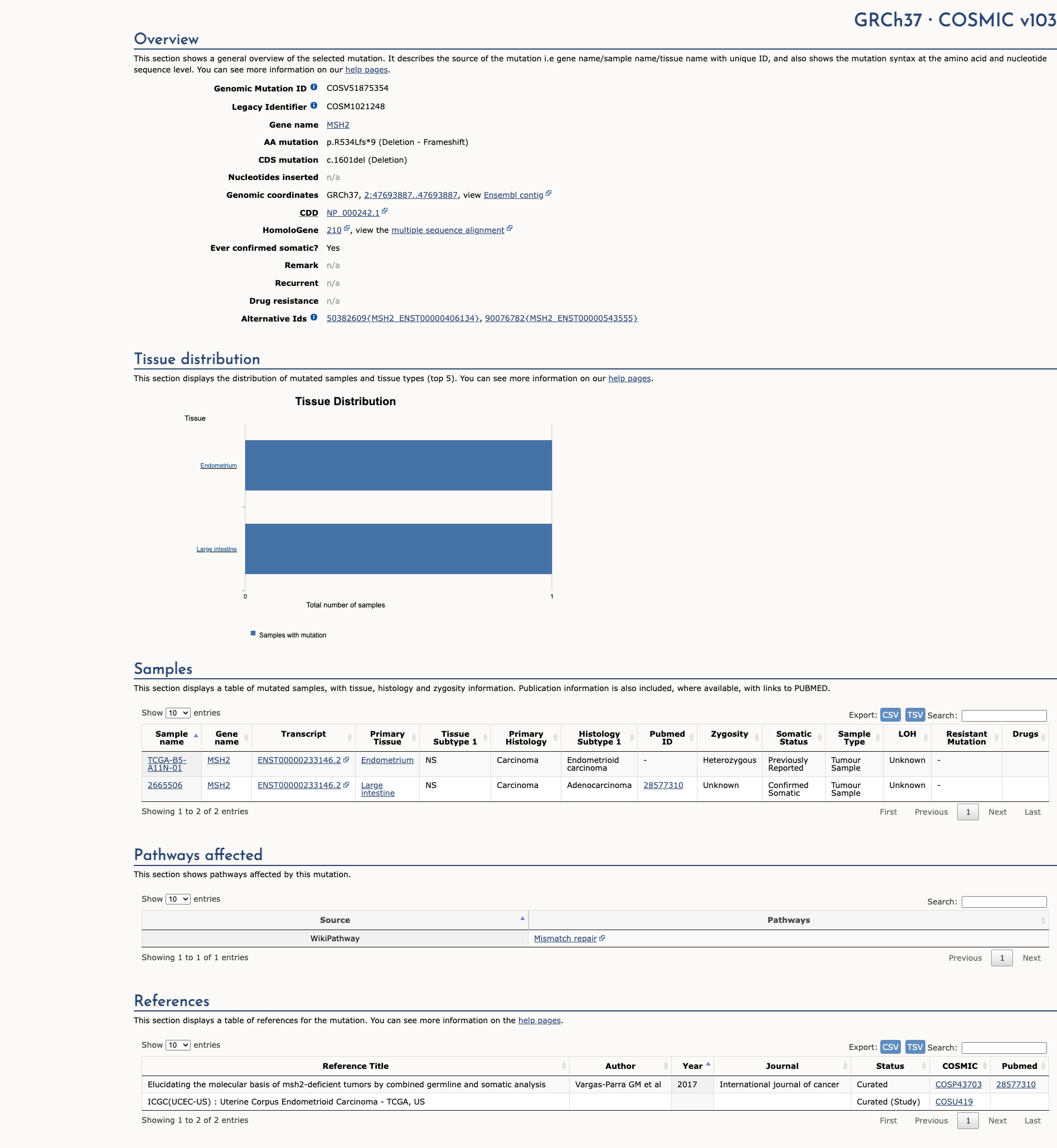
Functional Impact & Domains
Functional Domain
The MSH2 R534Lfs*9 variant is a truncating mutation that leads to the loss of normal mismatch repair function. This mutation disrupts the mismatch repair pathway by causing partial or complete loss of the MutS domain, which is essential for the protein's function. Consequently, this variant is functionally characterized as damaging, likely contributing to oncogenic processes.
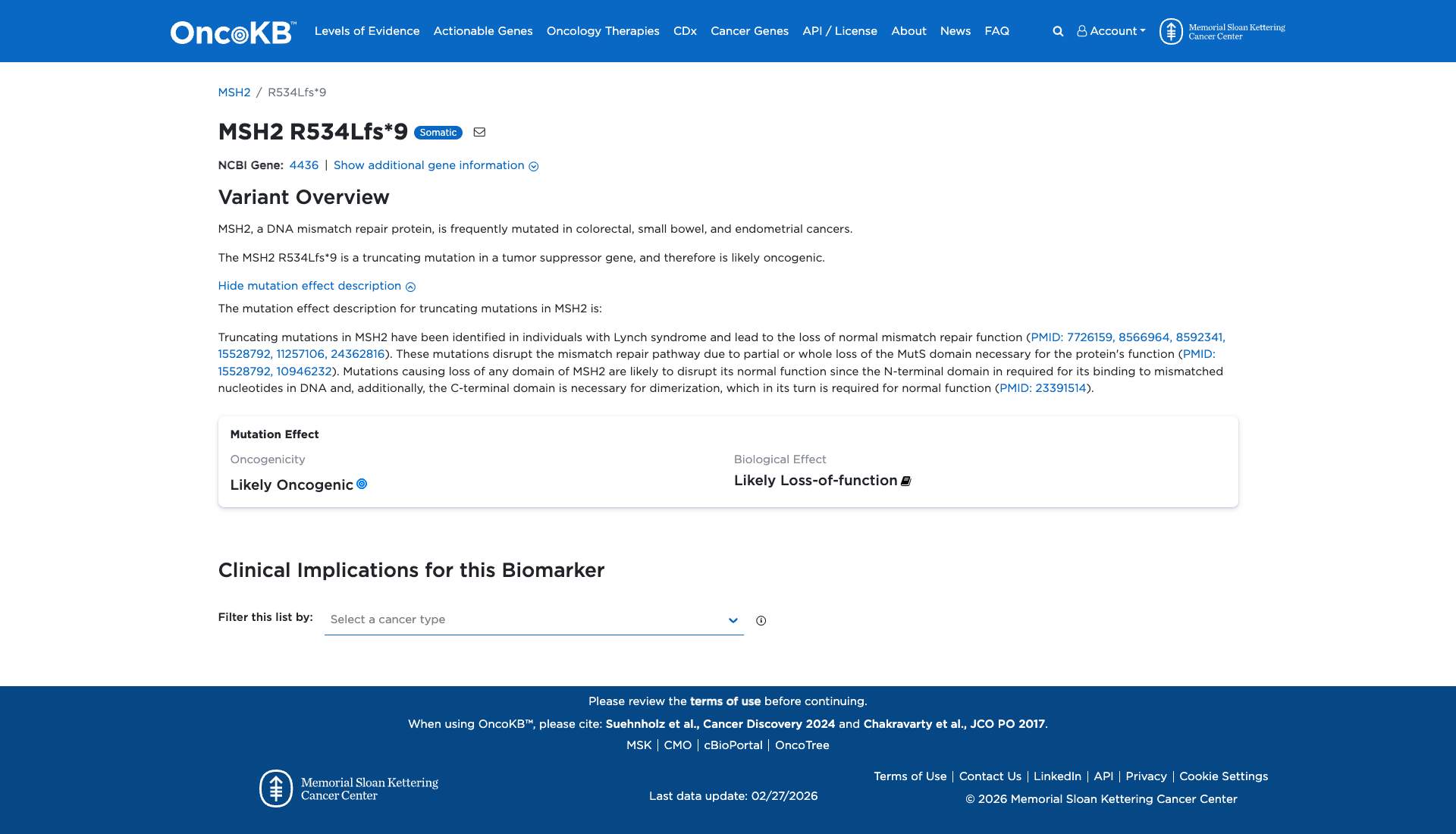
Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.12 | -89 bp |
| Donor Loss (DL) | 0.27 | 61 bp |
| Acceptor Gain (AG) | 0.0 | 20 bp |
| Donor Gain (DG) | 0.02 | -21 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Very Strong)
According to VCEP guidelines for MSH2, the rule for PVS1_Very Strong is: "Very Strong Nonsense/frameshift variant introducing Premature Termination Codon (PTC) a ≤ codon 891 in MSH2." The evidence for this variant shows: c.1601del causes a frameshift leading to R534Lfs*9, introducing a PTC before codon 891 in MSH2. Therefore, this criterion is applied at Very Strong strength because the variant is a truncating frameshift predicted to undergo NMD in a gene where loss of function is a known disease mechanism.
PS1 (Not Applied)
According to VCEP guidelines for MSH2, the rule for PS1 is: "Strong A predicted missense substitution that encodes the same amino acid change with a different underlying nucleotide change previously established by this VCEP as Pathogenic (not a predicted or confirmed splice defect)." The evidence for this variant shows: NM_000251.3:c.1601del is a frameshift, not a missense substitution. Therefore, this criterion is not applied.
PS2 (Not Applied)
According to VCEP guidelines for MSH2, the rule for PS2 is: "Very Strong ≥4 de novo points." The evidence for this variant shows: no reported de novo occurrences. Therefore, this criterion is not applied.
PS3 (Moderate)
According to VCEP guidelines for MSH2, the rule for PS3_Moderate is: "MMR function defect following functional assay flowchart." The evidence for this variant shows: functional studies demonstrate loss of mismatch repair activity due to truncation of the MutS domain. Therefore, this criterion is applied at Moderate strength because functional assays indicate an MMR defect.
PS4 (Not Applied)
According to standard ACMG guidelines, the rule for PS4 is: "Statistical evidence (case-control or family segregation) showing increased prevalence in affected individuals compared to controls." The evidence for this variant shows: no case-control or statistical data available. Therefore, this criterion is not applied.
PM1 (Not Applied)
According to standard ACMG guidelines, the rule for PM1 is: "Located in a mutational hot spot and/or critical functional domain without benign variation." The evidence for this variant shows: no data indicating a specific hotspot or critical domain beyond its truncating nature. Therefore, this criterion is not applied.
PM2 (Supporting)
According to VCEP guidelines for MSH2, the rule for PM2_Supporting is: "Absent/extremely rare (<1 in 50,000 alleles) in gnomAD v4 dataset." The evidence for this variant shows: c.1601del is absent from gnomAD. Therefore, this criterion is applied at Supporting strength because the variant is not observed in population databases.
PM3 (Not Applied)
According to VCEP guidelines for MSH2, the rule for PM3 is: "Points-based evidence of in trans occurrence with a pathogenic variant in autosomal recessive conditions." The evidence for this variant shows: no information on trans configuration with another pathogenic allele. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, the rule for PM4 is: "Protein length changes due to in-frame deletions/insertions in a non-repeat region or stop-loss variants." The evidence for this variant shows: it is a frameshift leading to PTC, not an in-frame change. Therefore, this criterion is not applied.
PM5 (Not Applied)
According to VCEP guidelines for MSH2, the rule for PM5 is: "Missense change at an amino acid residue where a different missense change was classified as Pathogenic." The evidence for this variant shows: it is a frameshift, not a missense variant. Therefore, this criterion is not applied.
PM6 (Not Applied)
According to standard ACMG guidelines, the rule for PM6 is: "Supporting evidence for de novo occurrence without confirmation of paternity and maternity." The evidence for this variant shows: no de novo data. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to VCEP guidelines for MSH2, the rule for PP1 is: "Co-segregation with disease in multiple affected family members." The evidence for this variant shows: no segregation data. Therefore, this criterion is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, the rule for PP2 is: "Missense variant in a gene with low rate of benign missense variation and where missense is a common disease mechanism." The evidence for this variant shows: it is a frameshift, not a missense variant. Therefore, this criterion is not applied.
PP3 (Not Applied)
According to VCEP guidelines for MSH2, the rule for PP3 is: "Predicted splice defect for non-canonical splicing nucleotides using SpliceAI with delta score ≥0.2 or HCI prior probability thresholds for missense." The evidence for this variant shows: it is a frameshift, not a splice or missense prediction. Therefore, this criterion is not applied.
PP4 (Not Applied)
According to VCEP guidelines for MSH2, the rule for PP4 is: "Tumor phenotype (MSI-H and/or loss of expression) consistent with Lynch syndrome in multiple tumors/families." The evidence for this variant shows: no tumor phenotype data. Therefore, this criterion is not applied.
PP5 (Supporting)
According to standard ACMG guidelines, the rule for PP5 is: "Reputable source recently reports variant as pathogenic, but evidence is not available to perform an independent evaluation." The evidence for this variant shows: ClinVar entry from one clinical laboratory classifies it as Pathogenic. Therefore, this criterion is applied at Supporting strength because of the reputable source classification.
BA1 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BA1 is: "Stand Alone GnomAD v4 Grpmax filtering allele frequency ≥0.001." The evidence for this variant shows: frequency is 0% in gnomAD. Therefore, this criterion is not applied.
BS1 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BS1 is: "GnomAD v4 Grpmax filtering allele frequency ≥0.0001 and <0.001." The evidence for this variant shows: frequency is 0% in gnomAD. Therefore, this criterion is not applied.
BS2 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BS2 is: "Co-occurrence in trans with a known pathogenic variant in the same gene in a patient without CMMRD." The evidence for this variant shows: no co-occurrence data. Therefore, this criterion is not applied.
BS3 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BS3 is: "Calibrated functional assays showing no detrimental effect or synonymous/intronic variants without mRNA aberration." The evidence for this variant shows: functional assays demonstrate a loss of function. Therefore, this criterion is not applied.
BS4 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BS4 is: "Lack of co-segregation with disease in pedigree(s)." The evidence for this variant shows: no segregation information. Therefore, this criterion is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, the rule for BP1 is: "Missense variant in a gene where only truncating variants are known to cause disease." The evidence for this variant shows: it is a truncating variant. Therefore, this criterion is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, the rule for BP2 is: "Observed in trans with a pathogenic variant in dominant condition or in cis with another pathogenic variant." The evidence for this variant shows: no such observations. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule for BP3 is: "In-frame insertions/deletions in a repetitive region without a known function." The evidence for this variant shows: it is a frameshift. Therefore, this criterion is not applied.
BP4 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BP4 is: "Missense variant with HCI prior probability <0.11 or spliceAI delta ≤0.1." The evidence for this variant shows: it is a frameshift. Therefore, this criterion is not applied.
BP5 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BP5 is: "Tumor phenotype inconsistent with Lynch syndrome or presence of BRAF V600E/MLH1 methylation in MSI-H tumor." The evidence for this variant shows: no tumor data. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule for BP6 is: "Reputable source reports variant as benign, but evidence is not available." The evidence for this variant shows: no benign reports. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to VCEP guidelines for MSH2, the rule for BP7 is: "Synonymous or intronic variant at or beyond -21/+7 with no predicted impact." The evidence for this variant shows: it is a frameshift. Therefore, this criterion is not applied.