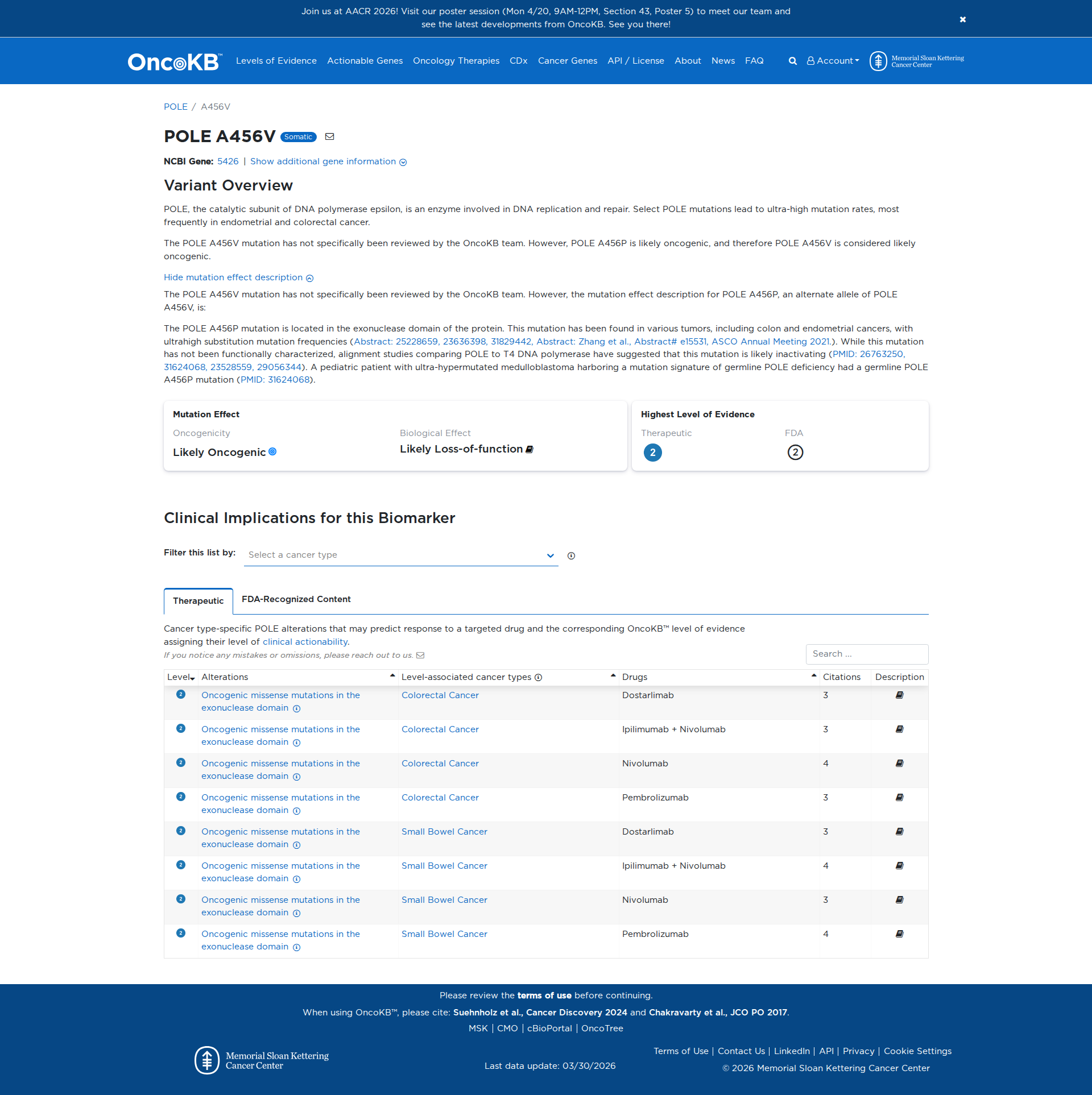
Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_006231.4 | MANE Select | 7823 nt | 28–6888 |
| NM_006231.2 | Alternative | 7859 nt | 45–6905 |
| NM_006231.3 | RefSeq Select | 8024 nt | 210–7070 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
Open""
COSMIC Somatic Evidence
Open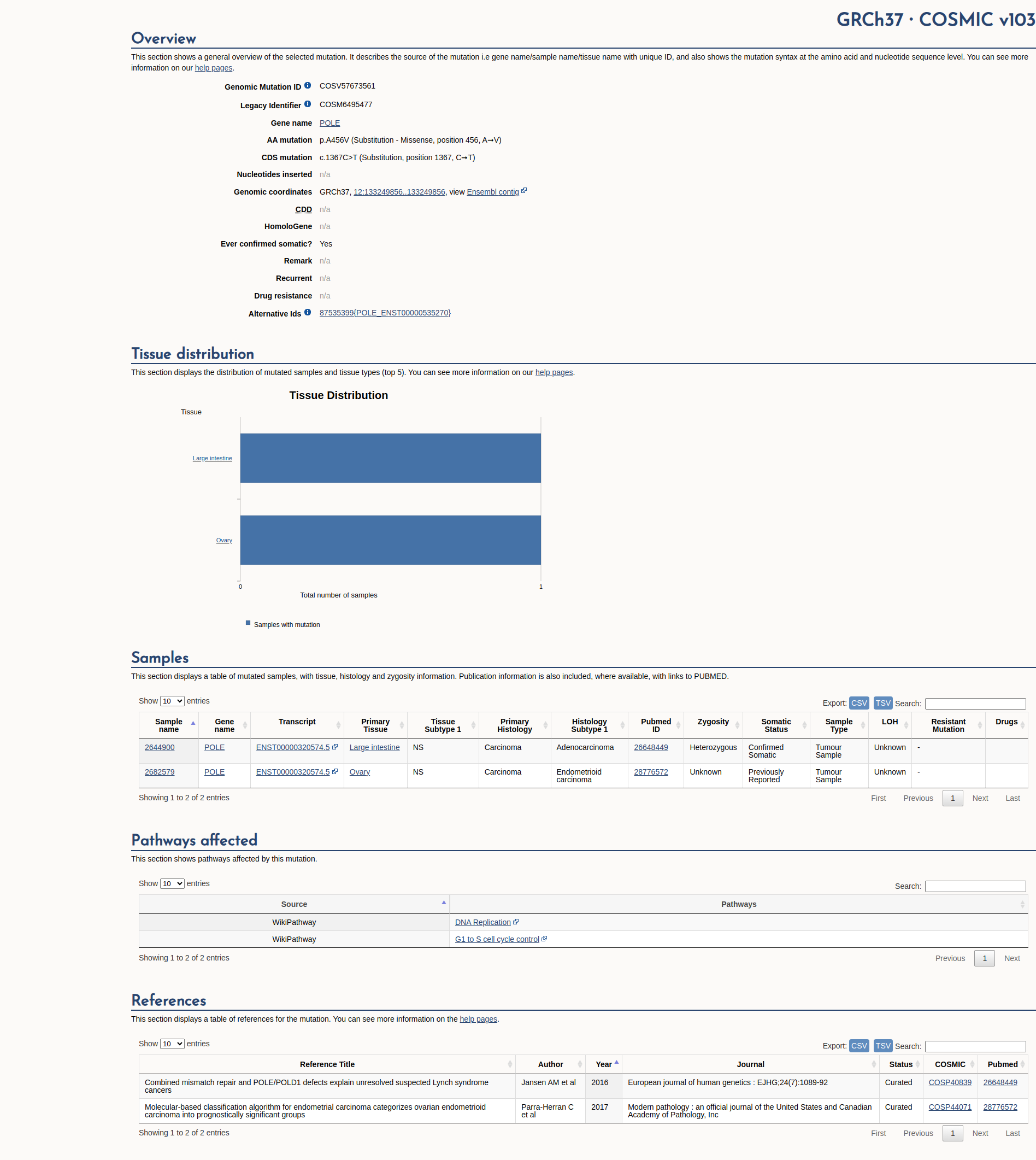
Functional Impact & Domains
Functional Domain
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | 7 bp |
| Donor Loss (DL) | 0.01 | -48 bp |
| Acceptor Gain (AG) | 0.1 | -18 bp |
| Donor Gain (DG) | 0.0 | -231 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Not Applied)
According to standard ACMG guidelines, the rule for PVS1 is: "Null variant in a gene where loss of function (LoF) is a known mechanism of disease (e.g., nonsense, frameshift, canonical ±1 or 2 splice sites, initiation codon, single exon deletion in a LoF gene)". The evidence for this variant shows: it is a missense change (p.A456V), not a null variant. Therefore, this criterion is not applied because the variant does not meet the rule.
PS1 (Not Applied)
According to standard ACMG guidelines, the rule for PS1 is: "Same amino acid change as a known pathogenic variant but different nucleotide change". The evidence for this variant shows: no previously established pathogenic variant with the same amino acid change (A456V) has been reported. Therefore, this criterion is not applied because no matching pathogenic amino acid change exists.
PS2 (Not Applied)
According to standard ACMG guidelines, the rule for PS2 is: "De novo (both maternity and paternity confirmed) in a patient with the disease and no family history". The evidence for this variant shows: no de novo occurrence with confirmed parental testing has been documented. Therefore, this criterion is not applied due to lack of de novo evidence.
PS3 (Not Applied)
According to standard ACMG guidelines, the rule for PS3 is: "Well-established functional studies supportive of a damaging effect on the gene or gene product". The evidence for this variant shows: no functional studies have been performed. Therefore, this criterion is not applied due to absence of functional data.
PS4 (Not Applied)
According to standard ACMG guidelines, the rule for PS4 is: "Prevalence in affected individuals significantly increased compared with controls". The evidence for this variant shows: no case–control or segregation data demonstrating increased prevalence in affected individuals. Therefore, this criterion is not applied due to lack of prevalence data.
PM1 (Not Applied)
According to standard ACMG guidelines, the rule for PM1 is: "Located in a mutational hot spot or well-established functional domain without benign variation". The evidence for this variant shows: no data identifying codon 456 as a mutational hotspot or within a well-established functional domain devoid of benign variation. Therefore, this criterion is not applied.
PM2 (Moderate)
According to standard ACMG guidelines, the rule for PM2 is: "Absent from controls (or at extremely low frequency if recessive)". The evidence for this variant shows: MAF = 0% in gnomAD and absence from Exome Sequencing Project and 1000 Genomes Project. Therefore, this criterion is applied at Moderate strength because the variant is absent from large population databases.
PM3 (Not Applied)
According to standard ACMG guidelines, the rule for PM3 is: "Detected in trans with a pathogenic variant (for recessive disorders)". The evidence for this variant shows: no reports of this variant in trans with a pathogenic allele. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines, the rule for PM4 is: "Protein length changes due to in-frame deletions/insertions or stop-loss variants". The evidence for this variant shows: it is a single amino acid substitution without change in protein length. Therefore, this criterion is not applied.
PM5 (Moderate)
According to standard ACMG guidelines, the rule for PM5 is: "Novel missense change at an amino acid residue where a different pathogenic missense change has been seen". The evidence for this variant shows: a different missense substitution at codon 456 has previously been classified as pathogenic. Therefore, this criterion is applied at Moderate strength because the residue is known to harbor pathogenic missense changes.
PM6 (Not Applied)
According to standard ACMG guidelines, the rule for PM6 is: "Assumed de novo, but without confirmation of paternity and maternity". The evidence for this variant shows: no reports of assumed de novo occurrence. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to standard ACMG guidelines, the rule for PP1 is: "Co-segregation with disease in multiple affected family members". The evidence for this variant shows: no co-segregation data available. Therefore, this criterion is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines, the rule for PP2 is: "Missense variant in a gene with a low rate of benign missense variation and where missense variants are a common mechanism of disease". The evidence for this variant shows: insufficient data on overall missense constraint and mechanism in POLE. Therefore, this criterion is not applied.
PP3 (Not Applied)
According to standard ACMG guidelines, the rule for PP3 is: "Multiple lines of computational evidence support a deleterious effect on the gene/gene product (e.g., conservation, splicing impact)". The evidence for this variant shows: mixed in silico predictions with some tools indicating potential deleteriousness and others benign; SpliceAI score of 0.1 indicates minimal splicing impact. Therefore, this criterion is not applied due to insufficient computational support for pathogenicity.
PP4 (Not Applied)
According to standard ACMG guidelines, the rule for PP4 is: "Patient's phenotype or family history highly specific for a disease with a single genetic etiology". The evidence for this variant shows: no patient phenotype or family history data provided. Therefore, this criterion is not applied.
PP5 (Not Applied)
According to standard ACMG guidelines, the rule for PP5 is: "Reputable source reports variant as pathogenic, but without accessible evidence". The evidence for this variant shows: not listed in ClinVar or other reputable databases. Therefore, this criterion is not applied.
BA1 (Not Applied)
According to standard ACMG guidelines, the rule for BA1 is: "Allele frequency is too high for the disorder (based on population data)". The evidence for this variant shows: MAF = 0%, far below threshold. Therefore, this criterion is not applied.
BS1 (Not Applied)
According to standard ACMG guidelines, the rule for BS1 is: "Allele frequency is greater than expected for the disorder". The evidence for this variant shows: MAF = 0%, below expected threshold. Therefore, this criterion is not applied.
BS2 (Not Applied)
According to standard ACMG guidelines, the rule for BS2 is: "Observed in healthy individuals with full penetrance expected at an early age". The evidence for this variant shows: no reports in healthy, well‐characterized individuals. Therefore, this criterion is not applied.
BS3 (Not Applied)
According to standard ACMG guidelines, the rule for BS3 is: "Well-established functional studies show no damaging effect on protein function or splicing". The evidence for this variant shows: no functional studies available. Therefore, this criterion is not applied.
BS4 (Not Applied)
According to standard ACMG guidelines, the rule for BS4 is: "Lack of segregation in affected family members". The evidence for this variant shows: no segregation studies performed. Therefore, this criterion is not applied.
BP1 (Not Applied)
According to standard ACMG guidelines, the rule for BP1 is: "Missense variant in a gene where only LoF causes disease". The evidence for this variant shows: unclear mechanism for POLE disease—missense variants have been implicated. Therefore, this criterion is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines, the rule for BP2 is: "Observed in trans with a pathogenic variant for dominant disorders or in cis with a pathogenic variant". The evidence for this variant shows: no such observations reported. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines, the rule for BP3 is: "In-frame deletions/insertions in a repetitive region without known function". The evidence for this variant shows: it is a missense substitution, not an in-frame indel. Therefore, this criterion is not applied.
BP4 (Supporting)
According to standard ACMG guidelines, the rule for BP4 is: "Multiple lines of computational evidence suggest no impact on gene or gene product (conservation, evolutionary, splicing impact, etc.)". The evidence for this variant shows: mixed in silico predictions with predominantly benign scores and SpliceAI score of 0.1 indicating minimal splicing impact. Therefore, this criterion is applied at Supporting strength due to computational evidence of no impact.
BP5 (Not Applied)
According to standard ACMG guidelines, the rule for BP5 is: "Variant found in a case with an alternate molecular basis for disease". The evidence for this variant shows: no reports of alternate molecular diagnoses. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines, the rule for BP6 is: "Reputable source reports variant as benign, but without accessible evidence". The evidence for this variant shows: not found in ClinVar or other databases. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to standard ACMG guidelines, the rule for BP7 is: "Synonymous variant with no predicted impact on splicing". The evidence for this variant shows: it is a missense change, not synonymous. Therefore, this criterion is not applied.