Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000059.4 | MANE Select | 11954 nt | 200–10456 |
| NM_000059.2 | Alternative | 11386 nt | 228–10484 |
| NM_000059.3 | RefSeq Select | 11386 nt | 228–10484 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThe c.2830A>T (p.Lys944Ter) variant identified in the BRCA2 gene of this individual leads to the premature termination of the protein at amino acid 944/3419 (coding exon 11/27). This variant is found with low frequency in gnomAD (3 heterozygotes, 0 homozygotes, allele frequency: 1.06e-5) suggesting it is not a common benign variant in the populations represented in these databases. This variant is reported as Pathogenic in ClinVar (VarID:51355) after review by the Expert Panel and has been reported in many affected individuals in the literature [PMID: 24737347; PMID: 23683081; PMID: 20104584; PMID: 28346442]. It is reported here as Pathogenic.
Variant summary: BRCA2 c.2830A>T (p.Lys944X) results in a premature termination codon, predicted to cause a truncation of the encoded protein or absence of the protein due to nonsense mediated decay, which are commonly known mechanisms for disease. Truncations downstream of this position have been classified as pathogenic by our laboratory. The variant allele was found at a frequency of 8e-06 in 250612 control chromosomes. c.2830A>T has been reported in the literature in multiple individuals affected with Hereditary Breast And Ovarian Cancer Syndrome (example, Rebbeck_2018). These data indicate that the variant is very likely to be associated with disease. Multiple clinical diagnostic laboratories and one expert panel (ENIGMA) have submitted clinical-significance assessments for this variant to ClinVar after 2014 without evidence for independent evaluation. All submitters classified the variant as pathogenic. Based on the evidence outlined above, the variant was classified as pathogenic.
The p.K944* pathogenic mutation (also known as c.2830A>T), located in coding exon 10 of the BRCA2 gene, results from an A to T substitution at nucleotide position 2830. This changes the amino acid from a lysine to a stop codon within coding exon 10. This mutation has been detected in multiple breast and ovarian cancer families to date (Mote PA et al. Genes Chromosomes Cancer, 2004 Mar;39:236-48; Capalbo C et al. Ann Oncol, 2006 Jun;17 Suppl 7:vii34-40; Thomassen M et al. Acta Oncol, 2008;47:772-7; Bodd TL et al. Acta Paediatr., 2010 Nov;99:1741-3; Heidemann S et al. Breast Cancer Res Treat, 2012 Aug;134:1229-39; Vietri MT et al. Clin. Chem. Lab. Med., 2013 Dec;51:2319-24; Lucas AL et al. Cancer, 2014 Jul;120:1960-7; Rebbeck TR et al. Breast Cancer Res, 2016 11;18:112; Susswein LR et al. Genet Med, 2016 08;18:823-32; Weren RD et al. Hum. Mutat., 2017 02;38:226-235; Heramb C et al. Hered Cancer Clin Pract, 2018 Jan;16:3; Wen WX et al. J Med Genet, 2018 02;55:97-103; Rebbeck TR et al. Hum Mutat, 2018 05;39:593-620; Carter NJ et al. Gynecol Oncol, 2018 12;151:481-488; Chen B et al. Aging (Albany NY), 2020 02;12:3140-3155; Lerner-Ellis J et al. J Cancer Res Clin Oncol, 2021 Mar;147:871-879). In one study, this variant was reported in 2/60,466 breast cancer cases and in 2/53,461 controls (Dorling et al. N Engl J Med. 2021 02;384:428-439). This mutation has also been reported in patients with pancreatic cancer (Lowery MA et al. J Natl Cancer Inst, 2018 10;110:1067-1074; Sorscher S et al. Oncologist, 2021 Jul). Of note, this alteration is also designated as 3058A>T in the literature. In addition to the clinical data presented in the literature, this alteration is expected to result in loss of function by premature protein truncation or nonsense-mediated mRNA decay. As such, this alteration is interpreted as a disease-causing mutation.
This submission and the accompanying classification are no longer maintained by the submitter. For more information on current observations and classification, please contact variantquestions@myriad.com.
This sequence change creates a premature translational stop signal (p.Lys944*) in the BRCA2 gene. It is expected to result in an absent or disrupted protein product. Loss-of-function variants in BRCA2 are known to be pathogenic (PMID: 20104584). This variant is present in population databases (rs80358533, gnomAD 0.004%). This premature translational stop signal has been observed in individual(s) with breast and/or ovarian cancer (PMID: 9150154, 20104584, 23683081, 26681312, 27836010). This variant is also known as 3058A>T. ClinVar contains an entry for this variant (Variation ID: 51355). For these reasons, this variant has been classified as Pathogenic.
"This variant has been reported in ClinVar as Pathogenic (37 clinical laboratories) and as pathogenic (1 clinical laboratories) and as Pathogenic by Evidence-based Network for the Interpretation of Germline Mutant Alleles (ENIGMA) expert panel."
COSMIC Somatic Evidence
Open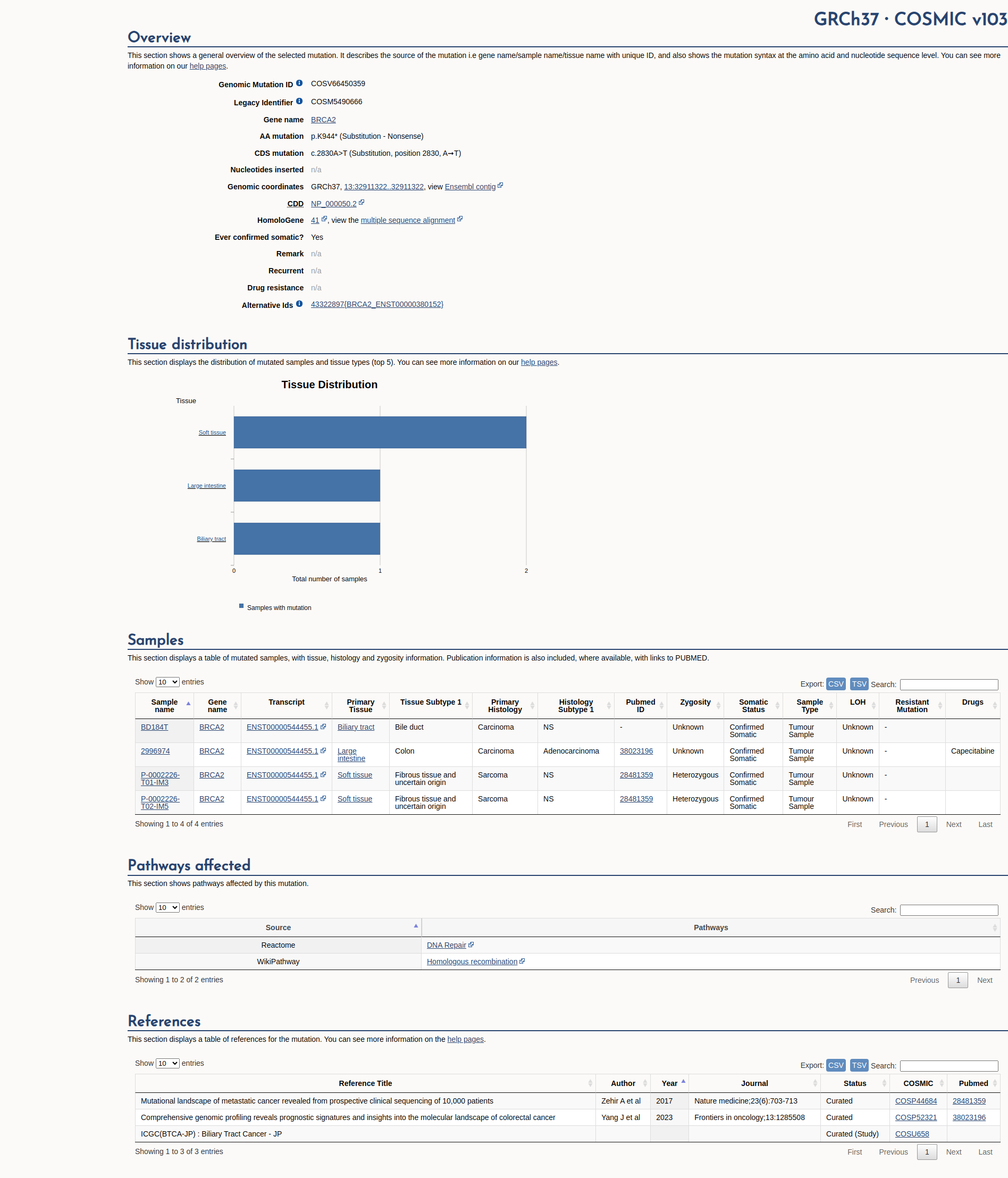
Functional Impact & Domains
Functional Domain
The BRCA2 K944* variant is a truncating mutation that results in the loss of critical protein domains, including the C-terminal DNA binding domain, nuclear localization signal, and CDK2 phosphorylation site. Experimental studies indicate that such truncating mutations impair the nuclear localization of BRCA2, which is essential for its normal function in maintaining homologous recombination during the DNA damage response. This functional disruption supports a damaging effect of the variant.

Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | 28 bp |
| Donor Loss (DL) | 0.0 | -432 bp |
| Acceptor Gain (AG) | 0.0 | -119 bp |
| Donor Gain (DG) | 0.0 | -30 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PVS1 (Very Strong)
According to VCEP guidelines: "Very Strong Null variant (nonsense, frameshift, splice site (donor/acceptor +/-1,2), initiation codon, single or multi-exon deletion) in a gene where loss of function (LOF) is a known mechanism of disease. Apply at appropriate strength according to PVS1 flowchart, which considers knowledge of clinically important functional domains. See Specifications Table 4 and Appendix D for details." The evidence for this variant shows: NM_000059.4:c.2830A>T introduces a premature stop codon (K944*), a truncating null variant in BRCA2, which is not in the last exon and is predicted to undergo nonsense-mediated decay. Therefore, this criterion is applied at Very Strong strength because it is a null variant in BRCA2, a gene where LOF is a known disease mechanism.
PS1 (Not Applied)
According to VCEP guidelines: PS1 applies to predicted missense substitutions where a previously classified pathogenic variant has the same amino acid change. The evidence for this variant shows: it is a nonsense variant (K944*), not a missense substitution. Therefore, this criterion is not applied because the variant does not meet the rule requirements.
PS2 (Not Applied)
According to standard ACMG guidelines: PS2 applies for confirmed de novo variants in a patient with the disease and no family history. The evidence for this variant shows: no de novo or parental testing data are available. Therefore, this criterion is not applied.
PS3 (Strong)
According to VCEP guidelines: "Strong Well-established in vitro or in vivo functional studies supportive of a damaging effect. Apply PS3 for assays measuring effect via protein only OR mRNA and protein combined. See Specifications Table 9 for code recommendations from calibrated published assays." The evidence for this variant shows: truncation of BRCA2 leading to loss of critical domains and impaired nuclear localization and homologous recombination in functional assays. Therefore, this criterion is applied at Strong strength because well-established functional studies demonstrate a damaging effect.
PS4 (Not Applied)
According to VCEP guidelines: PS4 applies when the prevalence of the variant in affected individuals is significantly increased compared to controls (case-control data with OR >=4 and p<=0.05). The evidence for this variant shows: no case-control or cohort data are provided. Therefore, this criterion is not applied.
PM1 (Not Applied)
According to VCEP guidelines: PM1 applies to variants located in a mutational hotspot or well-characterized functional domain without benign variation. The evidence for this variant shows: it is a truncating variant, not a missense or in-frame change in a defined hotspot domain. Therefore, this criterion is not applied.
PM2 (Supporting)
According to VCEP guidelines: "Supporting Absent from controls in an outbred population, from gnomAD v2.1 (non-cancer, exome only subset) and gnomAD v3.1 (non-cancer). Region around the variant must have an average read depth ≥25. See Appendix G for details." The evidence for this variant shows: extremely low allele frequency in gnomAD (MAF=0.00106%, 3/282020 alleles) and no homozygotes. Therefore, this criterion is applied at Supporting strength because the variant is absent or extremely rare in control populations.
PM3 (Not Applied)
According to VCEP guidelines: PM3 applies for biallelic variants in BRCA2 associated with Fanconi Anemia with scored points per proband. The evidence for this variant shows: no Fanconi Anemia phenotype or biallelic occurrence data. Therefore, this criterion is not applied.
PM4 (Not Applied)
According to standard ACMG guidelines: PM4 applies to protein length changes due to in-frame insertions/deletions or stop-loss variants. The evidence for this variant shows: it is a nonsense change causing truncation, not an in-frame indel or stop-loss. Therefore, this criterion is not applied.
PM5 (Not Applied)
According to VCEP guidelines: PM5_PTC applies to termination codon variants in an exon where different proven pathogenic PTCs have been seen. The evidence for this variant shows: exon location and other PTCs in this exon are not documented. Therefore, this criterion is not applied due to insufficient exon-level data.
PM6 (Not Applied)
According to standard ACMG guidelines: PM6 applies to assumed de novo variants without confirmation of parental status. The evidence for this variant shows: no de novo or parental testing information is available. Therefore, this criterion is not applied.
PP1 (Not Applied)
According to VCEP guidelines: PP1 applies for segregation in multiple affected family members quantified by Bayes score. The evidence for this variant shows: no family segregation data are provided. Therefore, this criterion is not applied.
PP2 (Not Applied)
According to standard ACMG guidelines: PP2 applies to missense variants in genes with low rate of benign missense variation. The evidence for this variant shows: it is a nonsense variant, not a missense. Therefore, this criterion is not applied.
PP3 (Not Applied)
According to VCEP guidelines: PP3 applies for missense/in-frame or splicing predictions with supportive in silico evidence. The evidence for this variant shows: it is a nonsense variant and in silico tools predict no splicing impact, but PP3 is not applicable to null variants. Therefore, this criterion is not applied.
PP4 (Not Applied)
According to VCEP guidelines: PP4 applies when the patient phenotype is highly specific and supports a single gene. The evidence for this variant shows: no individual phenotype data are provided. Therefore, this criterion is not applied.
PP5 (Supporting)
According to standard ACMG guidelines: "Supporting PP5: Reputable source recently reports variant as pathogenic, but the evidence is not available to the laboratory to perform an independent evaluation." The evidence for this variant shows: ClinVar entries from 37 labs and ENIGMA expert panel classify it as pathogenic. Therefore, this criterion is applied at Supporting strength because reputable sources report it as pathogenic without available data.
BA1 (Not Applied)
According to VCEP guidelines: BA1 applies when filter allele frequency is >0.1% in gnomAD. The evidence for this variant shows: MAF is 0.00106%, which is below the BA1 threshold. Therefore, this criterion is not applied.
BS1 (Not Applied)
According to VCEP guidelines: BS1 applies when filter allele frequency is >0.01%. The evidence for this variant shows: MAF is 0.00106%, below the BS1 threshold. Therefore, this criterion is not applied.
BS2 (Not Applied)
According to VCEP guidelines: BS2 applies in the absence of recessive disease features (Fanconi Anemia) with scored points per unaffected individuals. The evidence for this variant shows: no data on unaffected carriers or absence of FA phenotype. Therefore, this criterion is not applied.
BS3 (Not Applied)
According to VCEP guidelines: BS3 applies when well-established functional studies show no damaging effect. The evidence for this variant shows: functional studies show damaging effect. Therefore, this criterion is not applied.
BS4 (Not Applied)
According to VCEP guidelines: BS4 applies when lack of segregation is demonstrated quantitatively. The evidence for this variant shows: no segregation data. Therefore, this criterion is not applied.
BP1 (Not Applied)
According to VCEP guidelines: BP1 applies to silent or missense/in-frame variants outside critical domains with no splicing impact. The evidence for this variant shows: it is a nonsense variant. Therefore, this criterion is not applied.
BP2 (Not Applied)
According to standard ACMG guidelines: BP2 applies when a variant co-occurs with a pathogenic variant in trans or cis without phenotype. The evidence for this variant shows: no co-occurrence data. Therefore, this criterion is not applied.
BP3 (Not Applied)
According to standard ACMG guidelines: BP3 applies to in-frame indels in repetitive regions without functional impact. The evidence for this variant shows: it is a nonsense variant. Therefore, this criterion is not applied.
BP4 (Not Applied)
According to VCEP guidelines: BP4 applies for missense/in-frame or splicing predictions supporting a benign interpretation. The evidence for this variant shows: it is a nonsense variant; computational predictions do not apply. Therefore, this criterion is not applied.
BP5 (Not Applied)
According to standard ACMG guidelines: BP5 applies when a variant is observed in trans with a pathogenic variant in a disorder without specific phenotype. The evidence for this variant shows: no such co-observation. Therefore, this criterion is not applied.
BP6 (Not Applied)
According to standard ACMG guidelines: BP6 applies when a reputable source reports a variant as benign but evidence is unavailable. The evidence for this variant shows: no benign classifications from reputable sources. Therefore, this criterion is not applied.
BP7 (Not Applied)
According to VCEP guidelines: BP7 applies to silent or intronic variants with no splicing impact. The evidence for this variant shows: it is a nonsense variant. Therefore, this criterion is not applied.