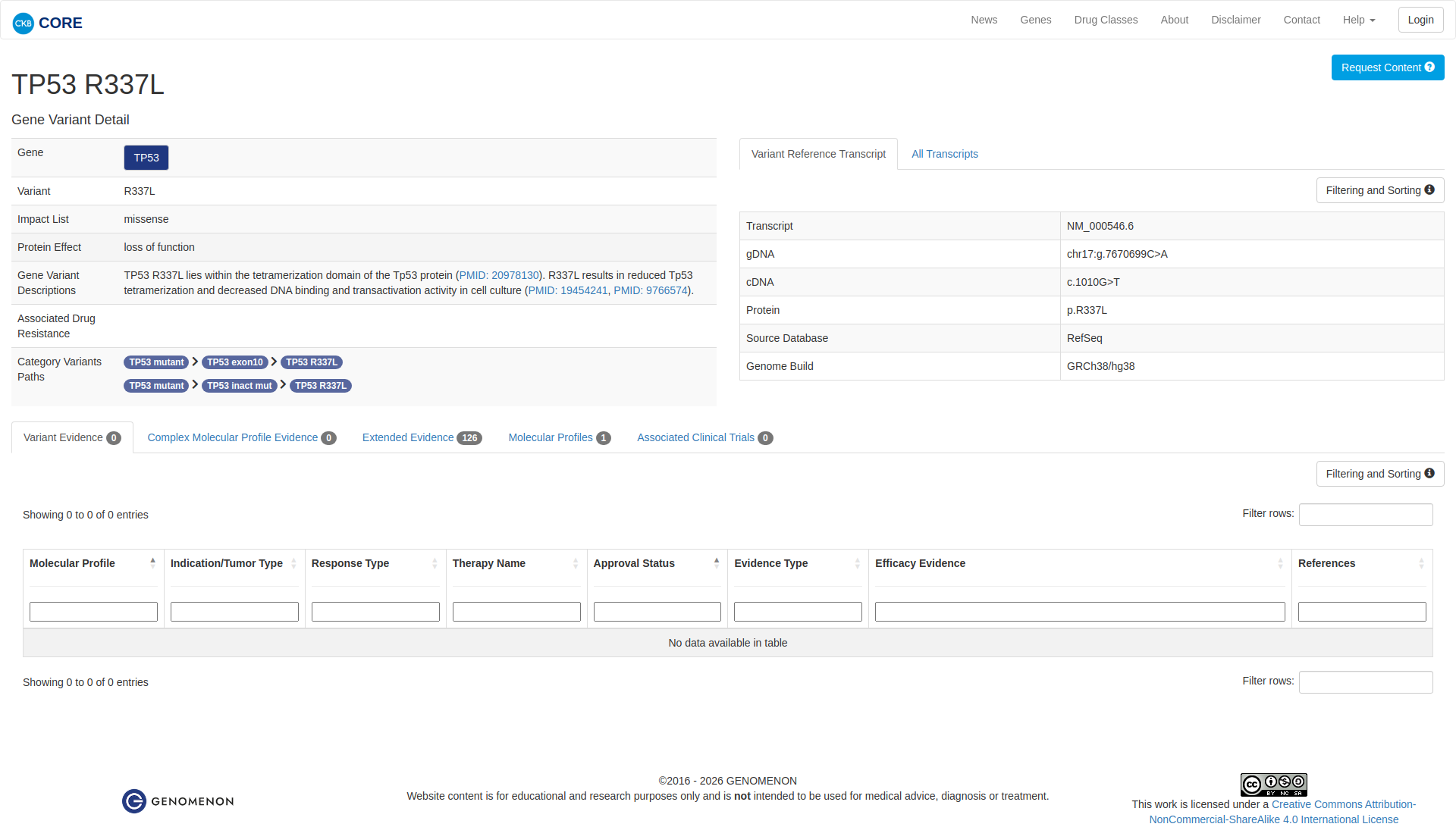
Genetic Information
Gene & Transcript Details
| ID | Status | Details |
|---|---|---|
| NM_000546.3 | Alternative | 2640 nt | 252–1433 |
| NM_000546.5 | RefSeq Select | 2591 nt | 203–1384 |
| NM_000546.6 | MANE Select | 2512 nt | 143–1324 |
| NM_000546.4 | Alternative | 2586 nt | 198–1379 |
| NM_000546.2 | Alternative | 2629 nt | 252–1433 |
Variant Details
Clinical & Population Data
Population Frequency
gnomADClinVar
OpenThe p.R337L pathogenic mutation (also known as c.1010G>T), located in coding exon 9 of the TP53 gene, results from a G to T substitution at nucleotide position 1010. The arginine at codon 337 is replaced by leucine, an amino acid with dissimilar properties. This alteration has been observed in at least one individual with a personal and/or family history that is consistent with TP53-related disease (Ambry internal data). Studies conducted in human cell lines indicate this alteration is deficient at growth suppression and has no dominant negative effect (Giacomelli AO et al. Nat Genet, 2018 Oct;50:1381-1387). This variant is reported to have non-functional transactivation in yeast-based assays (Kato S et al. Proc Natl Acad Sci U S A, 2003 Jul;100:8424-9). This variant is in the tetramerization domain of the TP53 protein and was not able to form tetramers (Kawaguchi T et al. Oncogene. 2005 Oct;24:6976-81). A thermal denaturation study predicted that several tetramerization domain mutants, including p.R337L, are thermally unstable at or near body temperature, and the authors note that earlier studies showed that p53 proteins with the p.R337L mutation exhibit an overall decrease in DNA-binding and transactivation activity (Kamada, R et al. J Biol Chem. 2011 Jan 7;286(1):252-8). This alteration has been observed numerous times as a somatic mutation in the cancerhotspots.org database (Chang MT et al. Cancer Discov. 2018 02;8:174-183). Based on internal structural analysis, this alteration is structurally deleterious (Ambry internal data). Other variant(s) at the same codon, p.R337H (c.1010G>A), have been identified in individual(s) with features consistent with Li-Fraumeni syndrome (Garritano et al. Hum Mutat. 2010 Feb;31(2):143-50; Ribeiro RC et al. Proc Natl Acad Sci U S A. 2001 Jul 31;98(16):9330-5; Giacomazzi J et al, PLoS ONE 2014 ; 9(6):e99893). This variant is considered to be rare based on population cohorts in the Genome Aggregation Database (gnomAD). This amino acid position is well conserved in available vertebrate species. In addition, the in silico prediction for this alteration is inconclusive. Based on the supporting evidence, this variant is interpreted as a disease-causing mutation.
This variant is considered likely pathogenic. Functional studies indicate this variant impacts protein function [PMID: 9766574, 16007150, 19454241, 9704930, 10653977, 9704931, 20978130]. This variant is expected to disrupt protein structure [Myriad internal data, PMID: 20978130].
"This variant has been reported in ClinVar as Pathogenic (1 clinical laboratories) and as Likely pathogenic (4 clinical laboratories) and as Likely Pathogenic by ClinGen TP53 Variant Curation Expert Panel, ClinGen expert panel."
COSMIC Somatic Evidence
Open
Functional Impact & Domains
Functional Domain
Error in OpenAI Consolidation. OncoKB: TP53R337LTP53R337LSomaticNCBI Gene:7157|Show additional gene information Variant OverviewTP53, a tumor suppressor in the DNA damage pathway, is the most frequently mutated gene in cancer.The TP53 R337L mutation is likely oncogenic.Hide mutation effect description The TP53 R337L mutation is located in the protein's oligomerization domain. This mutation has been found in the germlines of patients with Li-Fraumeni syndrome and adrenocortical carcinoma (PMID: 11481490). In vitro and structural studies have demonstrated that this mutation is likely inactivating, as evidenced by decreased transcriptional activity and protein stability in the mutant compared to wildtype, as well as the mutant's inability to form p53 tetramers, which is required for TP53 DNA binding activity (PMID: 19454241, 20978130). JAX-CKB: TP53 R337L lies within the tetramerization domain of the Tp53 protein (PMID: 20978130). R337L results in reduced Tp53 tetramerization and decreased DNA binding and transactivation activity in cell culture (PMID: 19454241, PMID: 9766574).

Click on previews to view full database entries. External databases may require institutional access.
Computational Analysis
Pathogenicity Predictions
SpliceAISpliceAI Scores
Window: ±500bp| Effect Type | Score | Position |
|---|---|---|
| Acceptor Loss (AL) | 0.0 | 60 bp |
| Donor Loss (DL) | 0.0 | 151 bp |
| Acceptor Gain (AG) | 0.0 | -289 bp |
| Donor Gain (DG) | 0.0 | -90 bp |
VCEP Guidelines
Applied ACMG/AMP Criteria (VCEP Specific)
PS3 (Unknown (Pre-LLM))
From pre-LLM assessment (LLM Failed)
PM2 (Unknown (Pre-LLM))
From pre-LLM assessment (LLM Failed)
PP5 (Unknown (Pre-LLM))
From pre-LLM assessment (LLM Failed)
BP4 (Unknown (Pre-LLM))
From pre-LLM assessment (LLM Failed)